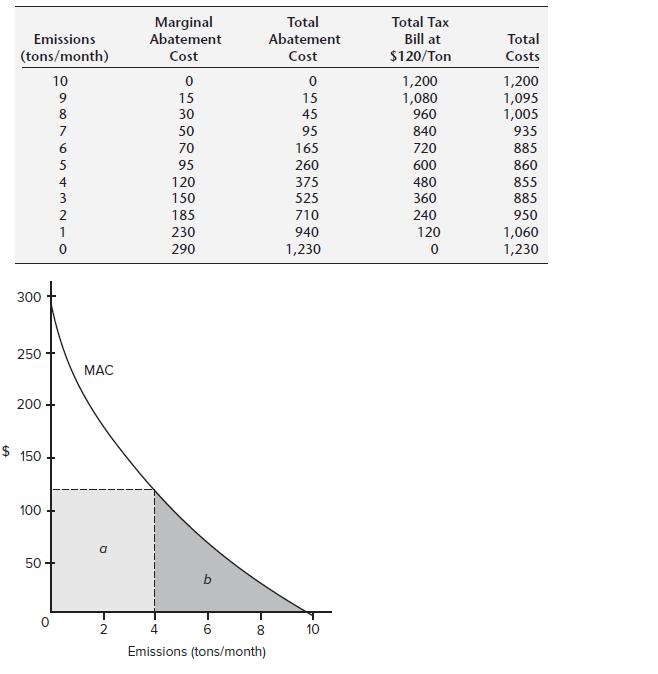
Refer to Figure 12.1. Suppose the emissions tax is $95/ton/month. Find the efficient level of emissions for
Question:
Refer to Figure 12.1. Suppose the emissions tax is $95/ton/month. Find the efficient level of emissions for the firm. Explain why the firm would not reduce its emissions to 1 ton/month.
Transcribed Image Text:
Emissions (tons/month) 10 300 250 200 $ 150 100 50 43312S000 9 8 7 6 5 MAC a Marginal Abatement Cost 0 15 30 50 70 95 120 150 185 230 290 b 4 6 Emissions (tons/month) 8 Total Abatement Cost 0 15 45 95 165 260 375 525 710 940 1,230 10 Total Tax Bill at $120/Ton 1,200 1,080 960 840 720 600 480 360 240 120 0 Total Costs 1,200 1,095 1,005 935 885 860 855 885 950 1,060 1,230
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
To find the efficient level of emissions for the firm we need to identify the point at which the mar...View the full answer

Answered By

Chandrasekhar Karri
I have tutored students in accounting at the high school and college levels. I have developed strong teaching methods, which allow me to effectively explain complex accounting concepts to students. Additionally, I am committed to helping students reach their academic goals and providing them with the necessary tools to succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Business questions
-
The profits associated with producing Q for Nash Enterprises are ( = 20Q - Q2, and marginal benefits are MB = 20 - 2Q. Pollution damages (costs) associated with its production are D = 10Q; the...
-
KYC's stock price can go up by 15 percent every year, or down by 10 percent. Both outcomes are equally likely. The risk free rate is 5 percent, and the current stock price of KYC is 100. (a) Price a...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Why are variances usually written off to cost of goods sold?
-
On December 31, 2012, Yates Co. prepared an adjusting entry for $12,000 of earned but unrecorded management fees. On January 16, 2013, Yates received $26,700 cash in management fees, which included...
-
In Problem 10.16, we projected financial statements for Walmart Stores, Inc. (Walmart) for Years +1 through +5. The data in Exhibits 12.17-12.19 (pages 948-950) include the actual amounts for 2012...
-
Your \(70-\mathrm{kg}\) friend always thinks he can dominate you in contests of strength because, even though you are a very athletic gymnast, your inertia is \(63 \mathrm{~kg}\). This time, while...
-
You are a manager at Glass Inc.??a mirror and window supplier. Recently, you conducted a study of the production process for your single-side encapsulated window. The results from the study are...
-
Develop a Java program to implement a neural network with backpropagation for a specific problem (e.g., XOR function approximation).
-
Emission charges are sometimes seen as creating a double burden: Firms must pay the costs of reducing emissions and also pay the government for polluting discharges. How might a charge system be...
-
When emission charges are put into effect, who ultimately ends up paying for them? Is this fair?
-
In Problems 3974, solve each system of equations using matrices (row operations). If the system has no solution, say that it is inconsistent. 3x + y = 2x = y + -y+ 4x + z = 2/31 7 = 1 z 2y 8 00 | 3
-
Describe government efforts to reduce audit failures.
-
Which GAAP require the use of depreciation for assets that have useful lives beyond 1 year? Explain why this is the case.
-
How do accountants keep track of the number of units sold if they are using the periodic inventory method?
-
What are the four basic cost flow methods for inventory valuation? What are the implications for financial reporting?
-
Explain the four steps in the depreciation process.
-
Using the heats of fusion and vaporization for water, calculate the change in enthalpy for the sublimation of water: H2O(s) H2O(g) Using the H value given in Exercise 24 and the number of hydrogen...
-
The production budget of Artest Company calls for 80,000 units to be produced. If it takes 30 minutes to make one unit and the direct labor rate is $16 per hour, what is the total budgeted direct...
-
Environmental protection programs are frequently designed to require all polluters to cut back emissions by a certain percentage. What are the perverse incentives built into this type of program?
-
Consider the example of Figure 11.3. Suppose we define as fair a cutback in which the two sources have the same total costs. Would an equiproportionate reduction be fair in this sense? A reduction...
-
For what types of pollution problems is voluntary action likely to be the most effective policy approach?
-
6) Find the determinant of the matrix 0 1 T10 51 -4 509
-
Determine if b is a linear combination of the vectors formed from the columns of the matrix A. 1 -4-4 A= 0 7 3-12 11 9 10 7
-
As a hard-working 30-year-old, it looks like I can scrape together $200 per month to put into the stock market. I hope to retire at age 70. If I can receive an 8% average return from now until age...

Study smarter with the SolutionInn App


