Lithium chloride, LiCl, has the same structure and therefore the same Madelung constant as NaCl. The equilibrium
Question:
Lithium chloride, LiCl, has the same structure and therefore the same Madelung constant as NaCl. The equilibrium separation in LiCl is 0.257 nm, and n = 7 in Equation 37.4. Find the ionic cohesive energy of the LiCl crystal.
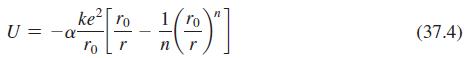
Transcribed Image Text:
ke ro U = -a ro Lr 1 (37.4)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 54% (11 reviews)
The ion ic cohesive energy of the Li Cl crystal is giv...View the full answer

Answered By

Rodrigo Louie Rey
I started tutoring in college and have been doing it for about eight years now. I enjoy it because I love to help others learn and expand their understanding of the world. I thoroughly enjoy the "ah-ha" moments that my students have. Interests I enjoy hiking, kayaking, and spending time with my family and friends. Ideal Study Location I prefer to tutor in a quiet place so that my students can focus on what they are learning.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Express the 7.84-eV ionic cohesive energy of NaCl in kilocalories per mole of ions.
-
Silicon is the fundamental component of integrated circuits. Si has the same structure as diamond. Is Si a molecular, metallic, ionic, or covalent-network solid?
-
Find the value of n?in Equation 38-32 that gives the measured dissociation energy of 741 kJ/mol for LiCl, which has the same structure as NaCl and for which r 0 = 0.257 nm. ke? (1-) U(r.) = -a- 38-32
-
Millennium Textiles Company makes silk banners and uses the weighted-average method of process costing. Direct materials are added at the beginning of the process, and conversion costs are added...
-
From which alkene could each of the following cyclopropane derivatives be prepared using the Simmons- Smith reaction? CH3
-
Design a low-pass filter with a cutoff frequency of \(50 \mathrm{krad} / \mathrm{s}\) and a passband gain of 200. Validate your design using Multisim. All \(R\) 's must be \(\geq 10 \mathrm{k}...
-
Hemlock Semiconductor Operations, LLC, and SolarWorld Industries Sachsen GmbH, are both companies involved in the manufacture of components for solar power products. Prior to the lawsuit, the two...
-
The adjusted trial balance columns of the worksheet for Alshwer Company, owned by M. Alshwer, are as follows. Instructions (a) Complete the worksheet by extending the balances to the financial...
-
RELATIONAL DATABASE CONCEPTS: Describe the basic steps required to install the Oracle, SQL Server, and MySQL relational database management systems (RDBMSs) and the major challenges that the user may...
-
On 1 June 20X7 Kha bought 60% of Sha paying $76,000 cash. The summarized statement of financial positions for the two entities as at 30 November 20X7 are Kha $ Sha $ Non-current assets Property,...
-
A salt crystal contains 10 21 sodiumchlorine pairs. How much energy would it take to compress the crystal to 90% of its normal size?
-
Youre researching the possibility of storing radioactive waste in underground salt formations. In support of this idea, youd like to demonstrate that salt is extremely resistant to compression. You...
-
What is the term for a more complex production strategy that combines approaches from more than one basis strategy?
-
Sheldon had salary income of $40,000. In addition, he had the following gains and losses on his property transactions. Long term capital gain = $14,000; short-term capital gain = $6,000; long-term...
-
In what manner did the leader's conduct exert significant influence, and what rationale supports this assessment?
-
An hourly employee is paid $30/hour for a 40 hour week. Overtime is paid at time-and-a-half for hours worked in excess of 40 hours. In the prior week, the employee worked 48 hours. Federal income...
-
answer these questions. 1. Define Multi-echelon Inventory Optimization (MEIO). 2. Discuss the leading drivers for the adoption of MEIO. 3. How does MEIO impact safety stocks? Within a supply chain...
-
(a) You are organising some large-scale fund-raising event for a lot of charities together, and managed to secure some major donors. Each donor agrees to donate some amount of money and each charity...
-
Many MNEs have greater ability to control and reduce their effective tax rates when expanding international operations. If Genedak-Hogan was able to reduce its consolidated effective tax rate from...
-
SCHEDULE OF COST OF GOODS MANUFACTURED The following information is supplied for Sanchez Welding and Manufacturing Company. Prepare a schedule of cost of goods manufactured for the year ended...
-
The plate has a width of 0.5 m. If the stress distribution at the support varies as shown, determine the force P applied to the plate and the distance d to where it is applied. 4 m- (15x2) MPa- 30 MPa
-
Determine the average normal stress in each of the 20-mm-diameter bars of the truss. Set P = 40 kN. 1.5 m 2 m
-
If the average normal stress in each of the 20-mm-diameter bars is not allowed to exceed 150 MPa, determine the maximum force P that can be applied to joint C. 1.5 m 2 m
-
For her dissertation, Catherine wanted to study the role of women in management positions in organisations. In particular, she was interested in difficulties women experience in management positions....
-
Hattersley Electrics is a division of the Hattersley Group PLC, a United Kingdom based manufacturing conglomerate whose main markets were, until recently, the high tech aerospace and the defence...
-
The Town Manager has asked you to organize a Local Emergency Action Planning Committee (LEAPC), Who do we Invite? Lets open the discussion up to who we might what to invite and why?

Study smarter with the SolutionInn App


