Lithium fluoride, LiF, has the same crystal structure as NaCl and therefore has essentially the same Madelung
Question:
Lithium fluoride, LiF, has the same crystal structure as NaCl and therefore has essentially the same Madelung constant a. Its ionic cohesive energy is -10.5 eV and the value of n in Equation 37.4 is 6.25 Find equilibrium ionic separation LiF.?
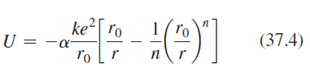
Transcribed Image Text:
ke ro U = (37.4) ľo [r
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 85% (7 reviews)
The equilibrium ionic separation in LiF can be determined using the Madelun...View the full answer

Answered By

Shahid Akbar
In my point of view, Education can't be finished when it's shared even it will increase in quantity. I like to share my knowledge to everyone who has the interest to improve his skills.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Determine the constant n in Equation 37.4 for potassium chloride (KCl), which has the same crystal structure as NaCl and for which r 0 = 0.315 nm and U 0 = -7.21 eV. ke ro 1 (37.4) U = ro [r
-
The theoretical density of germanium is 5.324 g/cm3 at 300 K. Germanium has the same crystal structure as diamond. What is the lattice constant of germanium at 300 K?
-
Find the value of n?in Equation 38-32 that gives the measured dissociation energy of 741 kJ/mol for LiCl, which has the same structure as NaCl and for which r 0 = 0.257 nm. ke? (1-) U(r.) = -a- 38-32
-
Describe the risk assessment approach used for Enterprise Risk Management(ERM) at Worker's Compensation Fund(WCF). How does this approach compare to ISO 31000? Does it consider risks with upside...
-
Design a preparation of each of the following compounds from an alcohol using sulfonate ester methodology- CH,CH,CH2 SCH
-
The sales manager wants to know whether salespeoples performance is related to their zodiac sign. Three hundred salespeople were surveyed. Their performance is summarized in the following table. Do...
-
A person donates a bag of clothes to Goodwill completely unaware that there is valuable sterling silver in the bag. The clothing and silver are subsequently sold, for a very economical price, to...
-
Andrew Thomas, a sandwich vendor at Hard Rock Cafes annual Rockfest, created a table of conditional values for the various alternatives (stocking decision) and states of nature (size of crowd): The...
-
The OLAP (Online Analytical Processing) discussion gives examples of multidimensional information. Give your own example of useful three-dimensional information.
-
Stanford Enterprises has provided its manufacturing estimated and actual data for the year end. The Controller has asked you to compute the predetermined overhead rate, the schedule of cost of goods...
-
Express the 7.84-eV ionic cohesive energy of NaCl in kilocalories per mole of ions.
-
Find the equilibrium ionic separation in LiF. 25. Find the wavelength of light emitted by a gallium phosphide (GaP) light-emitting diode. (See Table 37.1.) Table 37.1 Band-Gap Energies for Selected...
-
Plot each point given in polar coordinates, and find other polar coordinates of the point for which: (a) r > 0, -2 < 0 (b) r < 0, 0 < 2 (c) r > 0, 2 < 4 (5, 2/3)
-
For the simplex tableau X2 $1 52 P RHS 4 1 0 0 29 3 0 1 0 50 -4 0 01 0 perform one pivot operation and enter the resulting tableau below. The pivot element has a box around it. X X2 X 3 3 -10 $2 P RHS
-
Discuss some of the key principles of Quantum Physics, such as Heisenberg's Uncertainty principle, the concept of superposition, and the phenomenon of quantum tunneling. Question 4. Application of...
-
Define and explain the formation of polar and non-polar covalent bonds. State and discuss the cause of their similarities and differences. Use the case of water and hydrocarbons as the basis for your...
-
YAG laser operates at approximately 1.06 m wavelength and has gain with spectral width = 150 GHz. a) For a simple air-filled resonator based on parallel-plane mirrors spaced by 10 cm, how many...
-
What process does the "GU-AG Rule" apply to in eukaryotes. If the AG sequence was mutated in exon 1 in a gene with 3 exons, how would this process be affected?
-
Both domestic and international portfolio managers are asset allocators. What is their portfolio management objective?
-
ABC company leased new advanced computer equipment to STU Ltd on 1 January 2019.STULtd has to pay annual rental of $290,000 starting at 1 January 2019. It is a four years lease with ultimate rental...
-
The pump operates using the motor that has a power of 85 W. If the impeller at B is turning at 150 rev/min, determine the maximum shear stress in the 20-mm-diameter transmission shaft at A. 150...
-
The solid steel shaft AC has a diameter of 25 mm and is supported by smooth bearings at D and E. It is coupled to a motor at C, which delivers 3 kW of power to the shaft while it is turning at 50...
-
The motor delivers 50 hp while turning at a constant rate of 1350 rpm at A. Using the belt and pulley system this loading is delivered to the steel blower shaft BC. Determine to the nearest 1/8 in....
-
(5) For each of the following sets with a binary operation, determine if it a group or not and explain why. If it is not a group, you should provide at least one of the properties which is not...
-
Solve 1. f(x) = ln(2x-1) 2. f(x)=(x-1) 5x3+x 3. f(x) = e+x = 3 4. f(x) = e 5. f(x)=(3x -2x+1) 6. f(x)=2x-2x+3 7. f(x)= 8. f(x) = 1 2x-1 1 (4x + x)
-
4. Assume that country A's saving level is fixed at SA = 10 and country B's saving level is fixed at SB 25. Except the saving level, these two countries share the same economic parameters. In every...

Study smarter with the SolutionInn App


