Use the series expansion for e x (Appendix A) to show that Plancks law (Equation 34.3) reduces
Question:
Use the series expansion for ex (Appendix A) to show that Planck’s law (Equation 34.3) reduces to the Rayleigh–Jeans law (Equation 34.5) when l >> hc/kT.
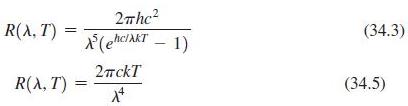
Transcribed Image Text:
2rhc? R(A, T) (34.3) (ehc/AKT - 1) 2nckT R(A, T) (34.5)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 72% (11 reviews)
According to Plancks law R T 2hc 2 5 e hckT 1 343 by using series expansion for e x we get R ...View the full answer

Answered By

User l_831638
Hello! My name is Sadhana.M.S. I made specilization in Nuclear Physics at Kuvempu university.I've been an physics tutor for degree level student . My motto is "teach student how to learn".
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A thin, uniform rod has length L and mass M. A small uniform sphere of mass m is placed a distance x from one end of the rod, along the axis of the rod (Fig. 12.34). (a) Calculate the gravitational...
-
(a) Verify that at high frequencies Wien's law is a good approximation to Planck's blackbody equation. (b) In June 1900 Rayleigh applied the equipartition theorem of classical statistical mechanics...
-
A Lens in a Liquid A lens obeys Snell's law, bending light rays at each surface an amount determined by the index of refraction of the lens and the index of the medium in which the lens is...
-
In a survey of 2230 U.S. adults, 1272 think that air travel is much more reliable than taking cruises. Construct a 95% confidence interval for the population proportion of U.S. adults who think that...
-
Show the 2p orbital's, and indicate the orbital overlap symbolized by the resonance structures for the carbocation in Eq. 15.32 on p. 711. Eq. 15.32 more important because each atom has a complete...
-
Determine the Geometrical Mean Radius (GMR) of the conductors with the arrangements shown in Fig. 3.3. The radius of each conductor is \(r\). 1) \(1.722 r\) 2) \(1.834 r\) 3) \(1.725 r\) 4) \(1.532...
-
An investment is guaranteed to have a unique value of IRR if which of the following is true? a. Alternating positive and negative cash flows b. An initial negative cash flow followed by all positive...
-
Samantha A. Cranston, age 37, is single and lives with her dependent mother at 426 Grouse Avenue, Allentown, PA 18105. Her Social Security number is 111-21-1113. 1. Samantha is a licensed...
-
Relational Databases are based on relational models and relational math. Write a brief description and history of the relational model.?
-
The Indian Spirit Company is operating with two divisions. Division H is producing a product line that is required as a component part of the product being manufactured by Division W. For Division H,...
-
An electron is moving at 10 6 m/s and you wish to measure its energy to an accuracy of 0.0. Whats the minimum time necessary for this measurement?
-
A photons wavelength is equal to the Compton wavelength of a particle with mass m. Show that the photons energy is equal to the particles rest energy.
-
Factor out the least power of the variable or variable expression. Assume all variables represent positive real numbers. 6r -2/3 - 5r -5/3
-
Applied Behavioral Analysts view behavior as occurring within a given context because individuals interact with their environments. Thus, behavior occurs in response to both individual and...
-
The Golden Eagle Corporation has the following items on their income and balance sheets (values in tables are in thousands: Balance Sheet Items (Assets) Last Year Two Years Ago Cash 520 500 Accounts...
-
Compare 1 mole of H 2, 1 mole of O 2 , and 1 mole of F 2 . Which has the largest number of molecules?
-
1. Tipateena Boutiques has the following equity accounts on its balance sheet: Common stock ($1.25 par, 500,000 shares) Contributed capital in excess of par Retained earnings Total common...
-
What questions did you ask the client to identify the safety issues that were presenting for them. Write the questions down or provide the mental health assessment form as per your workplace...
-
Sophia recently won a tax case litigated in the 7th Circuit. She recently heard that the Supreme Court denied the writ of certiorari. Should she be happy or not, and why?
-
The rate at which the temperature of an object changes is proportional to the difference between its own temperature and the temperature of the surrounding medium. Express this rate as a function of...
-
For the sequential reaction k A = 1.00 Ã 10 -3 s -1 . Using a computer spreadsheet program such as Excel, plot the concentration of each species for cases where k B = 10k A , k B = 1.5k A ,...
-
(Challenging) For the sequential reaction in Problem P35.19, plot the concentration of each species for the case where k B = k A . Can you use the analytical expression for [B] in this case?
-
For a type II second-order reaction, the reaction is 60% complete in 60 seconds when [A] 0 = 0.1 M and [B] 0 = 0.5 M. a. What is the rate constant for this reaction? b. Will the time for the reaction...
-
Evaluate the following limits. 3. lim f(x) if f(x) = 3 5x 2x, if x 1 4 (5 points) -3 1 + - 4x, if x < 1 x3 1 X - 1
-
Here are the height of 50 students who participated in a statistics survey: 242 23 34 60 45 66 77 23 34 45 674 54 52 54 65 76 89 89 76 56 76 34 43 33 47 56 58 266 16600 42 15 67 45 76 56 34 39 92 72...
-
Show that if f: [a, b] R is differentiable on (a, b) and f 0, then c = (a, b) such that f'(c) f(c) = + a-c 1 b-c Hint: Consider the function h(x) = f(x)(x - a)(x - b).

Study smarter with the SolutionInn App


