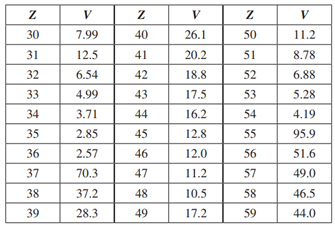
Using the table below, make a plot of atomic volume versus atomic number, for the elements from
Question:
Using the table below, make a plot of atomic volume versus atomic number, for the elements from Z = 30 to Z = 59 listed in the table. Comment on the structure of your graph in relation to the periodic table, the electronic structures of atoms, and their chemical properties. (Volumes are in units of 10-30 m3 .)
Transcribed Image Text:
V V V 30 7.99 40 26.1 50 11.2 31 12.5 41 20.2 51 8.78 32 6.54 42 18.8 52 6.88 33 4.99 43 17.5 53 5.28 34 3.71 44 16.2 54 4.19 35 2.85 45 12.8 55 95.9 36 2.57 46 12.0 56 51.6 37 70.3 47 11.2 57 49.0 38 37.2 48 10.5 58 46.5 39 28.3 49 17.2 59 44.0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
based on the data given in the table the graph of atomic volume versus atomic number for elements from Z 30 to Z 59 would display a generally decreasi...View the full answer

Answered By

Akshay Shete
I have extensive experience as a tutor, both online and in-person. I have worked with students of all ages and abilities, and am skilled at adapting my teaching style to meet the needs of each individual student. I have a strong background in a variety of subjects, including math, science, and English, and am able to break down complex concepts in a way that is easy for students to understand. In addition to my subject matter expertise, I am also a patient and supportive teacher, and am committed to helping my students succeed. Whether I am working with a struggling student who needs extra help to catch up, or an advanced student looking to get ahead, I am able to provide the guidance and support they need to reach their goals. Overall, my hands-on experience as a tutor has prepared me to be a confident and effective teacher, and I am excited to use my skills to help students succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Make a plot of bonding energy versus melting temperature for the metals listed in Table 2.3. Using this plot, approximate the bonding energy for copper, which has a melting temperature of 1084C.
-
In a laboratory experiment, air at 20C flows from a large tank through a 2- cm-diameter smooth pipe into a sea-level atmosphere, as in Fig. P6.149. The flow is metered by a long-radius nozzle of 1-cm...
-
A seven-component mixture is flashed at a specified temperature and pressure.(a) Using the K-values and feed composition given below, make a plot of the Rachford-Rice flash function at intervals of ?...
-
In the late 1980s, various states and the US Congress debated placing limits on sulfur emissions to reduce the impact of acid rain. Utilities that generated electricity using coal-powered plants felt...
-
In each of the following cases, imagine that the two reactants shown are allowed to reirct,in the presence of alcohol dehydrogenase. Tell whether the ethanol formed is chiral. If the ethanol is...
-
Briefly discuss the controls over accounts payable.
-
Purina entered in a contract with the defendant to sell the defendant piglets, known as weanlingsbaby pigs that have been weaned. It is uncontested that the buyer breached and that Purina is entitled...
-
Martin Shoes, Inc. manufactures and distributes orthopedic footwear. To sell its products, the marketing department requires sales personnel to call on the shoe retailers within their assigned...
-
Contemporary psychoanalysis has undergone a paradigm shift from drive reduction to the relational model. The most fundamental difference between relational and classical forms of psychoanalysis is...
-
Leigh, who is not a Circular 230 practitioner, is employed by Rose, a CPA. One of Roses clients has been notified that the IRS has selected his or her 2018 income tax return for audit. Rose had...
-
You work for a company that makes red helium?neon lasers widely used in physics experiments. Figure 36.19 shows an energy-level diagram for this laser. An electric current excites helium to a...
-
With sufficient energy, it?s possible to eject an electron from an inner atomic orbital. A higher-energy electron will then drop into the unoccupied state, emitting a photon with energy equal to the...
-
What are the responsibilities of the executor of an estate?
-
Most atoms happen to be about 1 Angstrom or 108 cm in radius, and typical nuclei are about 1013 cm in radius (actually, larger nuclei are somewhat larger, but we will ignore this complication)....
-
Capillary electrophoresis ( CE ) is notable for extremely narrow peak widths. What features of CE allow for these narrow peak widths? What are the advantages of narrow peaks?
-
Benfer Corporation's budgeA company's beginning income tax liability plus its budgeted income tax minus its budgeted income tax for the period equals its budgeted ending income tax liability ted...
-
How do different types of gene mutations influence genetic variation, disease susceptibility, and evolutionary processes? Provide examples
-
What are Coca-Cola's competitive advantages?
-
Sunrise Manufacturing, Inc, a U.S. multinational company, has the following debt components in its consolidated capital section. Sunrise's finance staff estimates their cost of equity to be 20%....
-
6. (Potential Energy and Conservation of Energy) What should be the spring constant k of a spring designed to bring a 1200-kg car to rest from a speed of 95 km/h so that the occupants undergo a...
-
The shear stressstrain diagram for an alloy is shown in the figure. If a bolt having a diameter of 0.25 in. is made of this material and used in the lap joint, determine the modulus of elasticity E...
-
The rubber block is subjected to an elongation of 0.03 in. along the x axis, and its vertical faces are given a tilt so that θ = 89.3°. Determine the strains ε x ,...
-
The lap joint is connected together using a 1.25 in. diameter bolt. If the bolt is made from a material having a shear stressstrain diagram that is approximated as shown, determine the permanent...
-
in a thousand words explain the theoretical differences between a corporate and a divisional cost of capital, and the potential for serious valuation errors if the incorrect WACC is used
-
You own an a townhome with an assessed value of $183,800. The tax rate is $2.20 per $100 of assessed value. (Round your answers to the nearest cent.)(a)What is the amount of property tax (in $)?$...
-
es Carter Paint Company has plants in four provinces. Sales last year were $100 million, and the balance sheet at year-end is similar in percent of sales to that of previous years (and this will...

Study smarter with the SolutionInn App


