With sufficient energy, it?s possible to eject an electron from an inner atomic orbital. A higher-energy electron
Question:
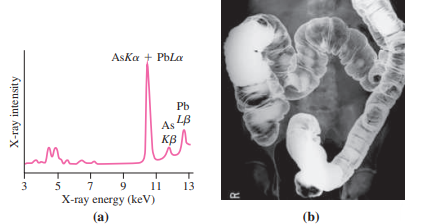
With sufficient energy, it?s possible to eject an electron from an inner atomic orbital. A higher-energy electron will then drop into the unoccupied state, emitting a photon with energy equal to the difference between the two levels. For inner-shell electrons, photon energies are in the keV range, putting them in the X-ray region of the spectrum. These characteristic X rays are labeled with the letter indicating the shell to which the electron drops, followed by a Greek letter indicating the higher level from which it drops; thus Ka designates a transition from the L shell to the K shell. Characteristic X rays provide scientists and physicians with an important diagnostic tool. Environmental scientists bombard pollution samples with high-energy electrons, knocking out inner-shell electrons and thus producing X-ray spectra that help identify contaminants (Fig. 36.20a). Geologists do the same with rocks. Medical radiologists reverse the process, exploiting the fact that X rays cause inner-shell transitions as well as complete ejection of inner-shell electrons. In particular, radiologists use the element barium in this way to produce high-contrast X-ray images of the intestinal tract (Fig. 36.20b).
?
In general, how should the energy of an element?s L? X rays compare with the energy of its K? X rays?a. They have less energy.b. They have the same energy.c. They have greater energy.d. You can?t tell without knowing the element.
Step by Step Answer:






