A 0.9826-g sample containing dimethyl phthalate, C 6 H 4 (COOCH 3 ) 2 (194.19 g/mol), and
Question:
A 0.9826-g sample containing dimethyl phthalate, C6H4(COOCH3)2 (194.19 g/mol), and unreactive species was refluxed with 50.00 mL of 0.1104 M NaOH to hydrolyze the ester groups (this process is called saponification).
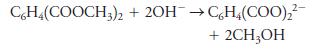
After the reaction was complete, the excess NaOH was back-titrated with 23.33 mL of 0.1597 M HCl. Calculate the percentage of dimethyl phthalate in the sample.
Transcribed Image Text:
C6H4(COOCH3)2 + 2OH →CH4(COO)₂²- + 2CH₂OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
Solution The percentage of dimethyl phthalate can be calculated using the following m...View the full answer

Answered By

Labindao Antoque
I graduated in 2018 with a Bachelor of Science degree in Psychology from Dalubhasaan ng Lungsod ng San Pablo. I tutored students in classes and out of classes. I use a variety of strategies to tutor students that include: lecture, discussions about the subject matter, problem solving examples using the principles of the subject matter being discussed in class , homework assignments that are directed towards reinforcing what we learn in class , and detailed practice problems help students to master a concept. I also do thorough research on Internet resources or textbooks so that I know what students need to learn in order to master what is being taught in class .
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Analytical Chemistry
ISBN: 9780357450390
10th Edition
Authors: Douglas A. Skoog, Donald M. West, F. James Holler, Stanley R. Crouch
Question Posted:
Students also viewed these Sciences questions
-
A 25.00-mL solution containing 0.03110 M Na2C2O4 was titrated with 0.02570 M Ca(NO3)2 to precipitate calcium oxalate:.Find pCa2+ at the following volumes of Ca(NO3)2: (a) 10.00; (b) Ve; (c) 35.00 mL....
-
A 50.0-mL sample containing Ni2+ was treated with 25.0 mL of 0.050 0 M EDTA to complex all the Ni2+ and leave excess EDTA in solution. The excess EDTA was then back titrated, requiring 5.00 mL of...
-
Calculate the volume in mL of a 1.420 M NaOH solution required to titrate the following solutions: (a) 25.00 mL of a 2.430 M HCl solution (b) 25.00 mL of a 4.500 M H2 SO4 solution (c) 25.00 mL of a...
-
Why does Erasmus attack Church officials in his In Praise of Folly? O For spending money on lavish art O For not allowing clergy to marry O For supporting military campaigns O For selling pardons and...
-
The mean ratio of students per teacher in U.S. public schools was 15.1 students per teacher in 2013, down 5.6% from 2000. What was the mean ratio of students per teacher in 2000?
-
Fortini Corporation had record sales in 2017. It began 2017 with an Accounts Receivable balance of $475,000 and an Allowance for Doubtful Accounts of $33,000. Fortini recognized credit sales during...
-
Describe the architecture of an expert system, and describe the roles of the various people involved in it.
-
Arantxa Corporation has outstanding 20,000 shares of $5 par value common stock. On August 1, 2010, Arantxa reacquired 200 shares at $80 per share. On November 1, Arantxa reissued the 200 shares at...
-
Ju(x,t) 1 au(x,t) = for-x < x < as two travelling waves when initial velocity is at Find the solution u(x, t) for the 1D wave equation 2 zero (make sure the first function represents wave travelling...
-
Cross State Trucking Brian Eagleton is a dispatch manager at Cross State Trucking, a major freight hauler based in Chicago, Illinois. Brian needs to develop a workbook that will summarize the drivin...
-
The Merck Index indicates that 10 mg of guanidine, CH 5 N 3 , may be administered for each kilogram of body mass in the treatment of myasthenia gravis. The nitrogen in a 4-tablet sample that had a...
-
Air was bubbled at a rate of 30.0 L/min through a trap containing 75 mL of 1% H 2 O 2 (H 2 O 2 + SO 2 H 2 SO 4 ). After 10.0 minutes, the H 2 SO 4 was titrated with 10.95 mL of 0.00242 M NaOH....
-
The displacement (in meters) of a particle moving in a straight line is given by the equation of motion s = 1/t2, where is measured in seconds. Find the velocity of the particle at times t = a, t -...
-
Beginning inventory Merchandise Finished goods Cost of merchandise purchased Cost of goods manufactured Ending inventory Merchandise Finished goods Unimart Bare Manufacturing $ 316,000 $ 632,000...
-
Castile Incorporated had a beginning balance of $2,100 in its Accounts Receivable account. The ending balance of Accounts Receivable was $2,800. During the period, Castile recognized $55,000 of...
-
Novak Inc. issued $3,840,000 of 11%, 10-year convertible bonds on June 1, 2025, at 99 plus accrued interest. The bonds were dated April 1, 2025, with interest payable April 1 and October 1. Bond...
-
Steph and Maya are standing atop a building looking down at a swimming pool. The pool is 10 m away from the building, and the building is 25 m tall. To jump off the pool, Maya tells Steph to run at 5...
-
The following are the transactions of Spotlighter, Incorporated, for the month of January. a. Borrowed $3,940 from a local bank on a note due in six months. b. Received $4,630 cash from investors and...
-
Panza Corporation experienced a fire on December 31, 2017, in which its financial records were partially destroyed. It has been able to salvage some of the records and has ascertained the following...
-
Differentiate the following terms/concepts: a. Personality types and money attitudes b. Planners and avoiders c. Moderating and adapting to biases d. "Perfectible judges" and "incorrigible judges"
-
Fill in the missing reagents below. Me OEt ?| Br Me SH OEt OMe OEt CN
-
Fill in the missing products below. excess HI Heat 1) Hg(OAc), Et 2) NABH, -: Na MCPBA 1) NaSH 2) H20 HBr
-
Propose a plausible synthesis for each transformation. a. b. c. d. e. f. g. h. i. j. k. l. m. n. o. p. q. r. s. t. u.
-
Dahlia Corporation has a current accounts receivable balance of $447,016. Credit sales for the year just ended were $4,950,605. a. What is the receivables turnover? Note: Do not round Intermediate...
-
Crenshaw Inc. is considering a change to activity-based product costing. The company produces two products, cell phones and tablet PCs, in a single production department. The production department is...
-
Alex Corporation reports the following components of stockholders' equity at December 31 of the prior year. Common stock-$25 par value, 50,000 shares authorized, 33,000 shares issued and outstanding...

Study smarter with the SolutionInn App


