Analysis of several plant-food preparations for potassium ion yielded the following data: The preparations were randomly drawn
Question:
Analysis of several plant-food preparations for potassium ion yielded the following data:
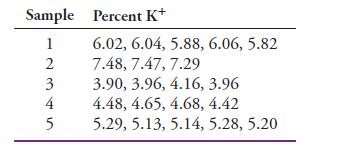
The preparations were randomly drawn from the same population.
(a) Find the mean and standard deviation s for each sample.
(b) Obtain the pooled value spooled.
(c) Why is spooled a better estimate of σ than the standard deviation from any one sample?
Transcribed Image Text:
Sample Percent K+ 6.02, 6.04, 5.88, 6.06, 5.82 7.48, 7.47, 7.29 3.90, 3.96, 4.16, 3.96 4.48, 4.65, 4.68, 4.42 5.29, 5.13, 5.14, 5.28, 5.20 1 N345
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Analytical Chemistry
ISBN: 9780357450390
10th Edition
Authors: Douglas A. Skoog, Donald M. West, F. James Holler, Stanley R. Crouch
Question Posted:
Students also viewed these Sciences questions
-
Find the mean and standard deviation of four-digit uniformly distributed lottery numbers (0000 through 9999).
-
For this data set, find the mean and standard deviation of the variable. The data represent the serum cholesterol levels of 30 individuals. Count the number of data values that fall within 2 standard...
-
For this data set, find the mean and standard deviation of the variable. The data represent the ages of 30 customers who ordered a product advertised on television. Count the number of data values...
-
Smart Price Company, a producer of black forest cakes, has budgeted sales and production (in units) for the last quarter in 2019 to be as follows: Sales Production October November 26,000 28,000...
-
Construct the complete sinking fund schedule. Calculate the total interest earned by adding up the interest earned column and by calculating the difference between the final balance in the fund and...
-
For each of the following linear transformations L: R2 R2, find a matrix representative, and then describe its effect on (i) The x-axis; (ii) The unit square S = (0 x, v 1): (iii) The unit disk D...
-
By modern standards, many injustices were committed on the path that today's wealthy nations took to power. Do these past injustices demand compensation by current-day people? Does history impose...
-
On January 1, Tellier Corporation had 60,000 shares of no-par common stock issued and outstanding. The stock has a stated value of $4 per share. During the year, the following transactions occurred....
-
Oriole Inc. sells prepaid telephone cards to customers in its convenience stores. When Oriole sells cards, it then pays the telecommunications company, Sheridan, for the value of the cards less a 20%...
-
Slacks.com received a bank statement from Cobb Bank indicating a bank balance of $7,700. Based on Slacks.coms check stubs, the ending checkbook balance was $8,136. Your task is to prepare a bank...
-
Before agreeing to the purchase of a large order of solvent, a company wants to see conclusive evidence that the mean value of a particular impurity is less than 1.0 ppb. What hypotheses should be...
-
Determination of phosphorous in blood serum gave results of 4.40, 4.42, 4.60, 4.48, and 4.50 ppm P. Determine whether the 4.60 ppm result is an outlier or should be retained at the 95% confidence...
-
Suppose the Environmental Protection Agency (EPA) would like to investigate the linear relationship between the engine size of sedans and the miles per gallon (MPG) they get. Data from the Excel file...
-
Afterglow.com Afterglow.com manufactures generic pharmaceutical pills in two departments, Mixing and Pill- making. Each pill contains 0.5 ounces of direct materials. Production information is as...
-
Jorge wants to start a CrossFit gym. He loves to work out, has rented a metal commercial building, and has rented a metal commercial building and has purchased the necessary equipment. He will have...
-
FIFO and LIFO Costs Under Perpetual Inventory System The following units of an item were available for sale during the year: Beginning inventory Sale First purchase Sale Second purchase Sale 30,000...
-
1.Describe the different types of weapons of mass destruction. 2. Local police play an integral role in preventing and responding terrorist attacks. How can local police agencies be prepared?
-
Helena Company manufactures and sells two products. Relevant per unit data concerning each product follow. Product Basic Deluxe Selling price $40 $52 Variable costs $22 $24 Machine hours 0.5 0.8 (a)...
-
Why does the "proper" operations strategy keep changing for companies that are world-class competitors?
-
Derive Eq. (18.33) from Eq. (18.32).
-
Calculate the molar solubility of SrC2O4 in a solution that has a fixed H3O+ concentration of (a) 1.0 10-6 M. (b) 1.0 10-9 M.
-
Calculate the molar solubility of BaSO4 in a solution in which [H3O+] is (a) 3.5 M. (b) 0.080 M.
-
Calculate the molar solubility of PbS in a solution in which [H3O+] is held constant at (a) 3.0 10-1 M and (b) 3.0 10-4 M.
-
How can the circular wait condition be detected and broken to prevent deadlock in complex systems with multiple interdependent resources ?
-
Case Study 1\ You have been appointed as a financial analyst for the Axis International Company, a profitable retail company. The director of Finance, belonging to the capital budgeting division has...
-
How can I find a rival hypothesis for my current thesis of Walmart's CEO should implement intra-team communication for all associates to help with communication barriers between managers and...

Study smarter with the SolutionInn App


