Calculate E 0 for the process Cu(NH3)42+ + Cu(NH3)2 + 2 NH; given that Cut + 2NH3Cu(NH3)2
Question:
Calculate E0 for the process
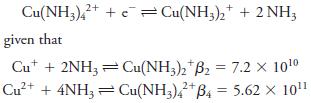
Transcribed Image Text:
Cu(NH3)42+ + Cu(NH3)2 + 2 NH; given that Cut + 2NH3Cu(NH3)2 B2 = 7.2 X 10¹⁰ 2+ Cu²+ + 4NH3 = Cu(NH3)42+ B4 = 5.62 × 10¹¹
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 87% (16 reviews)
E0 for the process CuNH3 2 CuNH 3 2 2 NH3 can be calculated using the standard red...View the full answer

Related Book For 

Fundamentals Of Analytical Chemistry
ISBN: 9780357450390
10th Edition
Authors: Douglas A. Skoog, Donald M. West, F. James Holler, Stanley R. Crouch
Question Posted:
Students also viewed these Sciences questions
-
(a) Calculate E0 for the process (b) Use the shorthand notation to describe a cell consisting of a saturated calomel reference electrode and a silver indicator electrode that could be used to measure...
-
Compute E0 for the process ZnY2- + 2e- Zn(s) + Y4- Where Y4- is the completely deprotonated anion of EDTA, The formation constant for ZnY2- is 3.2 1016.
-
The solubility product for Pb 3 (AsO 4 ) 2 is 4.1 x 10 -36 . Calculate E 0 for the reaction Pb(AsO4)2(s) + Ge = 3Pb(s) + 2AsO4-
-
Express the quantity of 3.225 kJ in calories.
-
X = + zs; x = 58.98, = 8.7, s = 2.6 (value of an observation)
-
Room registrations in the Toronto Towers Plaza Hotel have been recorded for the past 9 years. To project future occupancy, management would like to determine the mathematical trend of guest...
-
Shareholder value, as a concept, faces a number of problems. List these problems. Can any of the problems be overcome?
-
You have been assigned the task of evaluating two mutually exclusive projects with the following projected cash flows: If the appropriate discount rate on these projects is 10 percent, which would be...
-
Individually create an agenda for a professional meeting in which a Developmental Services Professional would participate and send your agenda in the format of a professional email. An Individual...
-
The subsidiaries of Perth International remit their earnings and investment proceeds to the Australian parent at the end of each year. The annual weighted average cost of capital or required rate of...
-
Briefly explain why the sparingly soluble product must be removed by filtration before you back-titrate the excess silver ion in the Volhard determination of (a) Chloride ion. (b) Cyanide ion. (c)...
-
Write chemical formulas for the following complex ions: (a) Hexamminezinc(II) (b) Dichloroargentate (c) Disulfatocuprate(II) (d) Trioxalatoferrate(III) (e) Hexacyanoferrate(II)
-
Why does the message receivers culture affect the choice of direct or indirect plan for communicating negative messages?
-
In this exercise you will see how matrix multiplication can be used to get a nice proof of the major identities that play a role in the extended Euclidean algorithm. We consider two integers a, b e Z...
-
Solve the following equation for "x" yx +6zx + 5yz = 0
-
Three charged particles are located at the corners of an equilateral triangle as shown in the figure below (let q = 3.20 C, and L= 0.910 m). Calculate the total electric force on the 7.00- C charge....
-
1. Write the equation of momentum for an inelastic collision of two cars, of mass1 and mass 2? 2. Write the two equations necessary for solving an elastic collision of two cars, of mass1 and mass 2?...
-
2. Using the distributed database described by the diagram below, specify the minimum types of operations the database must support to perform the following tasks. These operations include remote...
-
A financial analyst believes that the best way to predict a firms returns is by using the firms price-to-earnings ratio (PE) and its price-to sales ratio (PS) as explanatory variables. He estimates...
-
Which of the following is FALSE regarding the purchasing power parity (PPP). a. The PPP is a manifestation of the law of one price b. The PPP says that a country with a higher expected inflation can...
-
Suggest an efficient synthesis for each of the following transformations: a. b. c. d. Br Br
-
Identify the reagents necessary to achieve each of the following transformations: Br Br Br Br Br
-
Determine the configuration for every chirality center in each of the following compounds. a. b. c. HO H- - OH H,OH
-
ow do synergistic dynamics emerge within high-performance teams, and what role do diverse skill sets, complementary roles, and shared goals play in fostering collaborative innovation and collective...
-
Pietro Frozen Foods, Inc., produces frozen pizzas. For next year, Pietro predicts that 50,000 units will be produced, with the following total costs: Direct materials Direct labor Variable overhead...
-
You will complete two case studies over the course of the semester. These are to be completed individually. Each case study is found in the textbook and there are questions there to help guide your...

Study smarter with the SolutionInn App


