Suggest a range of sample masses for the indicated primary standard if it is desired to use
Question:
Suggest a range of sample masses for the indicated primary standard if it is desired to use between 35 and 45 mL of titrant:
(a) 0.180 M HClO4 titrated against Na2CO3(CO2 product).
(b) 0.102 M HCl titrated against Na2C2O4.
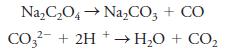
(c) 0.180 M NaOH titrated against benzoic acid.
(d) 0.090 M Ba(OH)2 titrated against KH(IO3)2.
(e) 0.065 M HClO4 titrated against TRIS.
(f) 0.060 M H2SO4 titrated against Na2B4O7 . 10H2O. Reaction:
![]()
Transcribed Image Text:
Na,C,O4→ Na,Co, + CO CO?- + 2H + → H2O + CO,
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (9 reviews)
a A suitable ...View the full answer

Answered By

Brian Otieno
I'm Brian , an experienced professional freelancer with countless hours of success in freelancing many subjects in different disciplines. Specifically, I have handled many subjects and excelled in many disciplines. I have worked on many Computer Science projects and have been able to achieve a lot in that field. Additionally, I have handled other disciplines like History, Humanities, Social Sciences, Political science, Health care and life science, and Religion / Theology. My experience generally in these subjects has made me able to deliver high-quality projects in a very timely fashion. I am very reliable at my job and will get the work done in time, no matter what. In Addition, I have managed to ensure that the work meets my client's expectations and does not cause an error. I am a hard-working and diligent person who is highly responsible for everything I do. Generally, Freelancing has made me more accountable for doing my job. Additionally, I have had a passion for writing for the last seven years in this field.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Analytical Chemistry
ISBN: 9780357450390
10th Edition
Authors: Douglas A. Skoog, Donald M. West, F. James Holler, Stanley R. Crouch
Question Posted:
Students also viewed these Sciences questions
-
It is desired to use a 0.75-m diameter beach ball to stop a drain in a swimming pool. Obtain an expression that relates the drain diameter D and the minimum water depth h for which the ball will...
-
It is desired to simulate flow past a ridge or bump by using a streamline above the flow over a cylinder, as shown in Fig. P8.50. The bump is to be a/2 high, as shown. What is the proper elevation h...
-
It is desired to produce an aligned and continuous fiber-reinforced epoxy composite having a maximum of 50 vol% fibers. In addition, a minimum longitudinal modulus of elasticity of 50 GPa (7.3 106...
-
The IQ and VIQ are tools insurance producers use to learn more about internal assumptions regarding factors that ultimately control policy performance. They have information in all of the following...
-
This problem demonstrates the dependence of an annuitys present value on the size of the periodic payment. Calculate the present value of 25 end-of-year payments of: a. $1000. b. $2000. c. $3000. Use...
-
CFA Corp. has a debt-equity ratio that is lower than the industry average, but its cash coverage ratio is also lower than the industry average. What might explain this seeming contradiction?
-
Identify four items that may be included in a representation letter.
-
In manufacturing its products, Trevano Corp. adds all direct material at the beginning of the production process. The companys direct labor and overhead are considered to be continuously at the same...
-
On December 31, 2024, capital balances of the partners in Michelle Charters are C. Anthony $64,800; M. Jason $50,400; and T. Michelle $36,000. The partners share profit in a 5:3:2 ratio,...
-
Montoure Company uses a periodic inventory system. It entered into the following calendar-year 2016 purchases and sales transactions. (For specific identification, units sold consist of 600 units...
-
A 25.0-mL aliquot of vinegar was diluted to 250 mL in a volumetric flask. Titration of 50.0-mL aliquots of the diluted solution required an average of 25.23 mL of 0.09041 M NaOH. Express the acidity...
-
The benzoic acid extracted from 86.7 g of catsup required a 15.61-mL titration with 0.0654 M NaOH. Express the results of this analysis in terms of percent sodium benzoate (144.10 g/mol).
-
Jason and Paula are married. They file a joint return for 2019 on which they report taxable income before the QBI deduction of $200,000. Jason operates a sole proprietorship, and Paula is a partner...
-
An executive summary, while written last, is the first part of a business plan with the gum stress company This document is what the executives will read to determine if the business plan has merit...
-
What do you find debatable in this reading assignment? Who would debate this passage and what arguments would he or she bring to the debate? What do you think about the possible positions one can...
-
A business plan template (Word File: BusinessPlanTemplate xx) and a Business Plan Guide (Wordfile: BusinessPlanGuide xx) which provides detailed instructions for the completion of each section of the...
-
explain the purpose of the business plan and its objectives. Be sure to include the following Purpose of the plan Three key elements of the business plan Objectives of a business plan
-
What do you find stimulating or exciting in this reading assignment? Summarize the section and explain why you find it stimulating.
-
Cheri's Beauty Products Ltd. reported the figures below at December 31, 2014, 2013, and 2012. The business has declared bankruptcy. You have been asked to review the business and explain why it...
-
Draw the appropriate control flow graph of the given pseudocode.Make sure to only use one number for blocks of code which are all sequential and when the first line is executed, all of those lines...
-
Calculate the 95% confidence interval for each set of data in Problem 7-4 if s is a good estimate of s and has a value of Set A, 0.30; Set C, 0.070; Set E, 0.0090
-
An atomic absorption method for the determination of the amount of iron present in used jet engine oil was found from pooling 30 triplicate analyses to have a standard deviation s = 3.6 mg Fe/mL. If...
-
How many replicate measurements are needed to decrease the 95 and 99% confidence limits for the analysis described in Problem 7-7 to 2.2 mg Fe / mL?
-
Pronghorn Corp reported net sales $675,000, cost of goods sold $438,750, operating expenses $154,000, and net income $69,525. Calculate the profit margin and gross profit rate. (Round Profit margin...
-
Victory Company uses weighted average process costing. The company has two production processes. Conversion cost is added evenly throughout each process. Direct ma added at the beginning of the first...
-
Here are selected 2022 transactions of Oriole Company. Jan. 1 June 30 Dec. 31 Retired a piece of machinery that was purchased on January 1, 2012. The machine cost $64,000 and had a useful life of 10...

Study smarter with the SolutionInn App


