The equilibrium constant for formation of CuCl 2 - is given by What is the solubility of
Question:
The equilibrium constant for formation of CuCl2- is given by
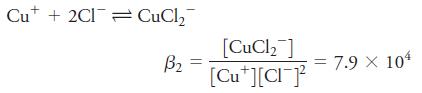
What is the solubility of CuCl in solutions having the following analytical NaCl concentrations:
(a) 5.0 M?
(b) 5.0 3 10-1 M?
(c) 5.0 3 10-2 M?
(d) 5.0 3 10-3 M?
(e) 5.0 3 10-4 M?
Transcribed Image Text:
Cu* + 2Cl= CuCl, [CuCl, ] B2 [Cu*][CI} = 7.9 X 104
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 55% (9 reviews)
Write equation for complex formation of CuCl 2 where 4 is the complex formation constant for CuCl 2 Cu 3 2Cl CuCl 2 2 CuCl 2 Cu 2 Cl 2 79 10 4 CuCl 2 ...View the full answer

Answered By

Umair Expert
Hi Everyone.
I have 6 years of teaching experience.
I am serving as a tutor 2 more websites.
I will provide you projects and solutions of questions related to any subject.
I am a good programmer.(PHP, Python)
I am good command in mathematics.
I am here to assist you.
I'll be very happy to work with you...
Thanks dear..
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Analytical Chemistry
ISBN: 9780357450390
10th Edition
Authors: Douglas A. Skoog, Donald M. West, F. James Holler, Stanley R. Crouch
Question Posted:
Students also viewed these Sciences questions
-
The equilibrium constant for the reaction H2 + at 1 atm and 1500C is given to be K. Of the reactions given below, all at 1500C, the reaction that has a different equilibrium constant is (a) H2 + 12O2...
-
The equilibrium constant for the H2 + ½ O2 H2O reaction at 1 atm and 1200 K is KP. Use this information to determine the equilibrium constant for the following reactions: (a) at l atm H, +...
-
The equilibrium constant for the reaction SO3 SO2 + O has the following values: Determine the average heat of dissociation using graphical method. T 800 K 900 K 1000 K 1105K | 0.0319 | 0.153 | 0.540...
-
Hrishi is a senior executive for a large manufacturing company in Mississauga Ontario where he has been employed for the past 10 years and his annual salary is $250,000 including bonus. He is 47...
-
Manuel bought a $100,000 bond with a 4% coupon for $92,300 when it had five years remaining to maturity. What was the prevailing market rate at the time Manuel purchased the bond? (Taken from CIFP...
-
When a Cobb-Douglas function with at least two inputs shows the existence of constant returns to scale, it implies that the marginal product of each input is diminishing. True or false? Explain.
-
Art offensive to religion, race, culture, or ethnicity has sometimes led to violence and death. Is the fear of violence a sufficient justification for artistic censorship? The statue of President...
-
See, I told you things would work out, said Barry Kresmier, president of Lomax Company. We expanded sales from $1.6 million to $2.0 million in 2009, nearly doubled our warehouse space, and ended the...
-
Sterling Optical andRoyal Optical both make glass frames and each is able to generateearnings before interest and taxes of $136,000. The separatecapital structures for Sterling and Royal are shown...
-
Clare Jaccard has $5,000 to invest. Because she is only 25 years old, she is not concerned about the length of the investments life. What she is sensitive to is the rate of return she will earn on...
-
Use activities to calculate the molar solubility of Zn(OH) 2 in (a) 0.0150 M KCl. (b) 0.0250 M K 2 SO 4 . (c) The solution that results when you mix 40.0 mL of 0.250 M KOH with 60.0 mL of 0.0250 M...
-
For a solution in which = 6.5 x 10 -2 , calculate K' sp for (a) AgSCN. (b) PbI 2 . (c) La(IO 3 ) 3 . (d) MgNH 4 PO 4 .
-
Tom and Lynda own Hercules Health Club. They provide the following information regarding expected membership. You also know that the monthly fee for an individual membership is $100 and the fee for a...
-
An art collector purchases a painting for $10,000. If the painting is expected to appreciate at an annual rate of 8%, what will be its estimated value in 10 years?
-
What is priming and how can it be studied? What symptoms do patients have who exhibit blindsight? Describe some of the evidence regarding the phenomena of priming and preconscious perception.
-
What are some real-world examples or case studies of companies effectively managing their cost of capital to enhance shareholder value or finance strategic initiatives?
-
What is a glycosidic linkage? What is the difference between alpha and beta glucose? Where do glyosidic linkages form? What is the difference between an alpha and a beta glyosidic linkage?
-
How can liquidity ratios, such as the current ratio and quick ratio, help assess a company's short-term financial health?
-
Green Corporation hires six individuals on January 4, 2016, all of whom qualify for the work opportunity credit. Three of these individuals receive wages of $8,500 during 2016, and each individual...
-
Uniform electric field in Figure a uniform electric field is directed out of the page within a circular region of radius R = 3.00 cm. The magnitude of the electric field is given by E = (4.50 x 10-3...
-
Calculate the theoretical cell potential of the following cells. If the cell is short-circuited, indicate the direction of the spontaneous cell reaction. (a) Zn ( Zn2+ (0.1000 M) (( Co2+ (5.87 ( 10-4...
-
Calculate the potential of the following two half-cells that are connected by a salt bridge: (a) A galvanic cell consisting of a lead electrode (right electrode) immersed in 0.0220 M Pb2+ and a zinc...
-
Write balanced net ionic equations to describe (a) The oxidation of Mn2+ to MnO4- by ammonium peroxydisulfate. (b) The oxidation of U4+ to UO22+ by H2O2. (c) The titration of H2O2 with KMnO4.
-
Speculate sales : Speculate on the projected sales. Justify your product or service by the numbers; discuss your projected revenue gain. Note: it MUST have an ROI that justifies the project for...
-
8.6 In Figure P8.6 the experimental unit step response of a process system is shown. (a) What is the transfer function that can be used to best describe the system dynamics among the following. 1 (1)...
-
An 84 year old female arrives to the ER today with her son with a complaint of new onset of confusion. What are your differential diagnoses for a patient with new altered mental status? Go through...

Study smarter with the SolutionInn App


