The following data are for a liquid chromatographic column: A chromatogram of a mixture of species A,
Question:
The following data are for a liquid chromatographic column:

A chromatogram of a mixture of species A, B, C, and D provided the following data:
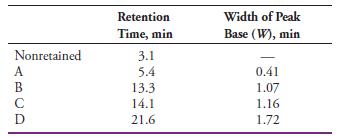
Calculate
(a) The number of plates from each peak.
(b) The mean and the standard deviation for N.
(c) The plate height for the column.
Transcribed Image Text:
Length of packing Flow rate 24.7 cm VM Vs 0.313 mL/min 1.37 ml 0.164 ml
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (12 reviews)
The following data from the chromatogram obtained for a mixture of the samples A B C and D Retention time min Width of peak base W min Nonretained 31 A 54 041 B 133 107 C 141 116 D 216 172 Here the Le...View the full answer

Answered By

Umair Expert
Hi Everyone.
I have 6 years of teaching experience.
I am serving as a tutor 2 more websites.
I will provide you projects and solutions of questions related to any subject.
I am a good programmer.(PHP, Python)
I am good command in mathematics.
I am here to assist you.
I'll be very happy to work with you...
Thanks dear..
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Analytical Chemistry
ISBN: 9780357450390
10th Edition
Authors: Douglas A. Skoog, Donald M. West, F. James Holler, Stanley R. Crouch
Question Posted:
Students also viewed these Sciences questions
-
The following data are for a simple steam power plant as shown in Fig. P6.99. State 1 2 3 4 5 6 7 P MPa 6.2 6.1 5.9 5.7 5.5 0.01 0.009 T C 45 175 500 490 40 h kJ/kg - 194 744 3426 3404 - 168 State 6...
-
The following data are for a simple steam power plant as shown in Fig. P6.99. State 6 has x6 = 0.92, and velocity of 600 ft/s. The rate of steam flow is 200 000 lbm/h, with 400 hp input to the pump....
-
The following data are for a simple steam power plant as shown in Fig. P6.39 State 1 2 3 4 5 6 7 P MPa 6.2 6.1 5.9 5.7 5.5 0.01 0.009 T C 45 175 500 490 40
-
In Canada, Canadian teachers and bosses and HIGH AUTHORITY FIGURES are not FEARED but RESPECTED. In CANADA TEACHERS, bosses and AUTHORITY FIGURES encourage feedback, OPINIONS and ENGAGE in discuss....
-
A conditional sale contract between Classic Furniture and the purchaser of a dining room set requires month-end payments of $250 for 15 months. Classic Furniture sold the contract to Household...
-
Here are the cash-flow forecasts for two mutually exclusive projects: a. Which project would you choose if the opportunity cost of capital is 2%? b. Which would you choose if the opportunity cost of...
-
You are engaged in doing the audit of a corporation whose records have not previously been audited by you. The corporation has both an independent transfer agent and a registrar for its capital...
-
The following chart was prepared at the beginning of a HRM (Human Resource Management) crash hiring project. The project begins with two activities: Assemble interview team (A) and Budget resources...
-
Discuss the importance of a company being seen by its public as "being ethical" in its marketing practices. What can a company do to ensure its marketers are practicing ethical marketing?
-
Hackers may be able to fool you into believing that their web site is actually a web site (such as a bank or credit card web site) that you trust. This may be done by misleading email, or even by...
-
Describe the preparation of exactly 1.00 L of 0.1000 M HCl from primary-standard-grade NaCl using a cation-exchange resin.
-
To determine the equilibrium constant for the reaction 25.0 mL of a 0.0100 M aqueous solution of I 2 was extracted with 10.0 mL of CHCl 3 . After extraction, spectrophotometric measurements revealed...
-
Two forces, F 1 and F 2 , are given by the vectors F 1 = (4i 5j) N and F 2 = (pi + qj) N. The resultant force, R = F 1 + F 2 acts in a direction which is parallel to the vector (3i j) a. Find the...
-
1- Describe one job that once existed but today is obsolete (or is slowly becoming obsolete) because of technology. 2- Define an Enterprise system and describe the uses and benefits of at least 2...
-
You reviewed the payroll records and noticed three employees were not paid the correct superannuation guarantee percentage. These employees were only paid 6% superannuation of their earnings base....
-
Hunt Company enters all cash payments in the cash payments journal. On February 21, the company issued Check No. 220 to Austin Company for merchandise purchased on January 29 for $6,000, terms 2/10,...
-
What gerontological nursing competencies seem to be absent from the practice of the nurse's nurse Haley describes?What are some of the factors that could have contributed to the nurses behaviors? How...
-
The value (V, in dollars) of a milling machine depends on the number of hours (x) it has been operated, as given by the formula V = 800,000-300x. After one year, the value of the machine is between...
-
Riley Kilgo Inc. purchased inventory costing $100,000 and sold 80% of the goods for $240,000. All purchases and sales were on account. Kilgo later collected 20% of the accounts receivable. 1....
-
A fuel pump sends gasoline from a car's fuel tank to the engine at a rate of 5.88 10-2 kg/s. The density of the gasoline is 735 kg/m3, and the radius of the fuel line is 3.18 10-3 m. What is the...
-
Dilute NaOH is introduced into a solution that is 0.050 M in Cu2+ and 0.040 M in Mn2+. (a) Which hydroxide precipitates first? (b) What OH- concentration is needed to initiate precipitation of the...
-
Silver ion is being considered for separating I- from SCN2 in a solution that is 0.040 M in KI and 0.080 M in NaSCN. (a) What Ag1 concentration is needed to lower the I2 concentration to 1.0 10-6 M?...
-
What mass of AgBr dissolves in 200 mL of 0.200 M NaCN? Ag+ + 2CN--Ag(CN), 2 = 1.3 1021
-
CHEM 3711/8711 Pre-Workshop Questions. Oxidation (Week 8) 1. For each of the compounds below indicate: a) b) whether you would expect them to be easily oxidised; and what would be the expected...
-
After the success of the company's first two months, Santana Rey continues to operate Business Solutions. The November 30, 2021, unadjusted trial balance of Business Solutions (reflecting its...
-
STAR Co. provides paper to smaller companies whose volumes are not large enough to warrant dealing directly with the paper mill. STAR receives 100-feet-wide paper rolls from the mill and cuts the...

Study smarter with the SolutionInn App


