The following data were obtained by gas-liquid chromatography on a 40-cm packed column: Calculate (a) An average
Question:
The following data were obtained by gas-liquid chromatography on a 40-cm packed column:
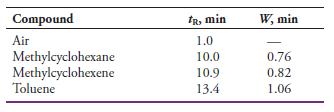
Calculate
(a) An average number of plates from the data.
(b) The standard deviation for the average in (a).
(c) An average plate height for the column.
Transcribed Image Text:
Compound IR, min W, min Air 1.0 Methylcyclohexane Methylcyclohexene Toluene 10.0 0.76 10.9 0.82 13.4 1.06
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)
The number of plates N for a chromatograph is calculated as N 554 x t ...View the full answer

Answered By

Manmohan V
First of all, I have devoted five years to gain the knowledge of chemistry bottom-up at my University. After my undergraduate, I started teaching and tutoring as well on some online platforms. I have worked as a Subject Metter Expert with three companies and gained perfection in Microsoft Office, Latex, ChemDraw and many online tools to deliver fast and accurate solutions.
As a freelancer, I am working on two online portals, where I have solved over 4000 queries in the past two years with a perfect quality score.
With my Creative skills of explanation, I guarantee that you will not face any problems with the solutions provided by me.
Thanks.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Analytical Chemistry
ISBN: 9780357450390
10th Edition
Authors: Douglas A. Skoog, Donald M. West, F. James Holler, Stanley R. Crouch
Question Posted:
Students also viewed these Sciences questions
-
In a recent survey, the following data were obtained in response to the question, If the number of summer classes were increased, would you be more likely to enroll in one or more of them? If a...
-
The following data were obtained when a cold-worked metal was annealed. (a) Estimate the recovery, recrystallization, and grain growth temperatures; (b) Recommend a suitable temperature for a...
-
Suppose that the following data were obtained by an investigator studying the influence of estrogen injections on change in the pulse rate of adolescent chimpanzees: a. What are the factors in this...
-
Which type of Organizational Model put the MOST emphasis on establishing MORALE and AUTONOMY O a. Hierarchical style Model b. O c. O d. Flat Management (Holocracy) Model Multi-divisional style Model...
-
A mortgage broker offers to sell you a mortgage loan contract that will pay $800 at the end of each month for the next 3 1/2 years, at which time the principal balance of $45,572 is due and...
-
Slush Corporation has two bonds outstanding, each with a face value of $2 million. Bond A is secured on the company's head office building; bond B is unsecured. Slush has suffered a severe downturn...
-
The Jones Company, located in Chicago, has been your client for many years. The company manufactures light machinery and has a calendar year closing. At December 31, 19X1 and 19X0 the following items...
-
Presented below is information related to equipment owned by Pujols Company at December 31, 2012. Cost (residual value $0) .........$9,000,000 Accumulated depreciation to date ....1,000,000...
-
Southeast U\'s campus book store sells course packs for $15.00each, the variable cost per pack is $12.00, fixed costs for thisoperation are $300,000, and annual sales are 95,000 packs. The...
-
Bear Park Ltd., a camping ground in the Lake District, has compiled the following financial information as of December 31, 2017. Instructions (a) Determine Bear Park's net income for 2017. (b)...
-
An open tubular column used for gas chromatography had an inside diameter of 0.15 mm. A volumetric flow rate of 0.85 mL/min was used. Find the linear flow velocity in cm/s at the column outlet.
-
Describe the preparation of exactly 1.00 L of 0.1000 M HCl from primary-standard-grade NaCl using a cation-exchange resin.
-
More than a million adult videos are rented each week. Nor, as the propagandists would have you believe, does viewing such material lead to violent sex crimes. If it did, there would be over one...
-
On the date of a formation of a joint venture, a venturer contributes non-monetary assets and receives an interest in the joint venture. How is the investment in the joint venture recorded?
-
What do you find interesting in this reading assignment? Summarize the point of interest and then discuss why you find it interesting.
-
. How is the formula for the volume of a sphere derived, and what is it?
-
Jones Inc. and Jeffrey Inc. form a jointly owned partnership, called JJ. Under the partnership agreement, both Jones and Jeffrey have joint control over the joint venture and have rights to the...
-
Why would corporate clients be interested in online technology training for their internal employees? How would they benefit from paying Pluralsight for training?
-
Deitrick Guitar Company is nearing the end of its worst year ever. With three weeks until year-end, it appears that net income for the year will have decreased by 20% from the previous year. Jim...
-
Ashlee, Hiroki, Kate, and Albee LLC each own a 25 percent interest in Tally Industries LLC, which generates annual gross receipts of over $10 million. Ashlee, Hiroki, and Kate manage the business,...
-
Neglecting any effects caused by volume changes, would you expect the ionic strength to (1) increase, (2) decrease, or (3) remain essentially unchanged when NaOH is added to a dilute solution of (a)...
-
Explain why the activity coefficient for dissolved ions in water is usually less than that for water itself.
-
Explain why the initial slope for Ca2+ in Figure 10-3 is steeper than that for K+? 0.8 0.6 0.4 0.2 Fe(CN)64- 0 0 0.1 0.2 0.3 0.4
-
Managers use CVP concepts to perform sensitivity analysis. Sensitivity analysis is a "what-if" technique that asks what will happen to a company's breakeven or target profit if sales price, costs, or...
-
John Fleming, chief administrator for Valley View Hospital, is concerned about the costs for tests in the hospital s lab. Charges for lab tests are consistently higher at Valley View than at other...
-
A company is looking at new equipment with an installed cost of $436,859. This cost will be depreciated straight-line to zero over the project's 6-year life, at the end of which the equipment can be...

Study smarter with the SolutionInn App


