The indicator HIn has an acid dissociation constant of 4.80 x 10 -6 at ordinary temperatures. The
Question:
The indicator HIn has an acid dissociation constant of 4.80 x 10-6 at ordinary temperatures. The accompanying absorbance data are for 8.00 x 10-5 M solutions of the indicator measured in 1.00-cm cells in strongly acidic and strongly alkaline media:
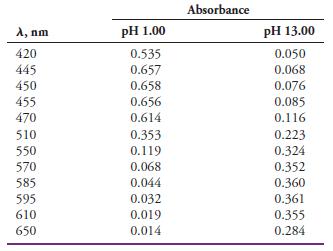
Estimate the wavelength at which absorption by the indicator becomes independent of pH (that is, the isosbestic point).
Transcribed Image Text:
Absorbance A, nm pH 1.00 pH 13.00 420 0.535 0.050 0.068 445 0.657 450 0.658 0.076 455 0.656 0.085 0.116 470 0.614 510 0.353 0.223 550 0.119 0.324 570 0.068 0.352 585 0.044 595 610 0.360 0.361 0.355 0.032 0.019 0.014 650 0.284
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (10 reviews)

Answered By

Surojit Das
I have vast knowledge in the field of Mathematics, Business Management and Marketing. Besides, I have been teaching on the topics Management leadership, Business Administration, Human Resource Management, Business Communication, Accounting, Auditing, Organizer Behaviours, Business Writing, Essay Writing, Copy Writing, Blog Writing since 2020. It is my personality to act quickly in any emergency situations when students need my services. I am very professional and serious in every questions students asked me at the time of dealing any projects. I have been serving detailed, quality, properly analysed research paper through the years.
4.80+
91+ Reviews
278+ Question Solved
Related Book For 

Fundamentals Of Analytical Chemistry
ISBN: 9780357450390
10th Edition
Authors: Douglas A. Skoog, Donald M. West, F. James Holler, Stanley R. Crouch
Question Posted:
Students also viewed these Sciences questions
-
The ionization constant Ka of an indicator HIn is 1.0 1026. The color of the nonionized form is red and that of the ionized form is yellow. What is the color of this indicator in a solution whose pH...
-
The acid dissociation constant for loss of a proton from cyclohexanol is 1 Ã 10-16 a. Draw an energy diagram for loss of a proton from cyclohexanol. b. Draw the contributing resonance...
-
The isoelectric point of an amino acid is the pH at which the molecule has no net charge. For glycine, that would be the pH at which virtually all glycine molecules are in the form +H3NCH2CO2-. If we...
-
Both high-income and low-income employees are covered by cafeteria plans. Under such plans, all employees may select from a list of non-taxable fringe benefits or they may elect to receive cash in...
-
Herb has made contributions of $2000 to his RRSP at the end of every six months for the past eight years. The plan has earned 9.5% compounded semiannually. He has just moved the funds to another plan...
-
Suppose the firm in Problem 3 had 35,000 shares of common stock outstanding. What is the earnings per share, or EPS, figure? What is the dividends per share figure?
-
As auditor for the Court Company, you decide to use variables sampling to estimate the total cost of an inventory of 1,150 items which has a net book value of \(\$ 260,400\). The auditor decides to...
-
Tanzanite Corporation issued $500,000 of 7% debentures to yield 11%, receiving $424,624. Interest is payable semiannually, and the bonds mature in five years. 1. What entries would be made by...
-
Consider the following two spot rates: CHF/USD 1.2500 and USD/EUR 1.2500 (a) What is the spot rate of CHF/EUR? (2 marks) (b) If HSBC gives a quote of CHF/EUR 1.5500, is there an arbitrage...
-
There are 2 shinobis with chakra levels 5 and 10 respectively and the desired sum of chakra levels is utmost 15 Starting with ke0, suy of chakra levels after attack max(5-0,0) + max(10- 0,0) 5+10 15....
-
Solutions of P and Q individually obey Beers law over a large concentration range. Spectral data for these species in 1.00-cm cells are (a) Plot an absorption spectrum for a solution that is 6.45 x...
-
Define (a) Transducer. (b) Photocurrent. (c) n-type semiconductor. (d) Majority carrier. (e) Depletion layer. (f) Dynodes in a photomultiplier tube.
-
In problem, find the center and radius of each circle. Graph each circle. Find the intercepts, if any, of each circle. x 2 + y 2 + 4x - 4y - 1 = 0
-
Transformation of the Banking Industry by Technology "Banks are intensive users of both IT and financial technologies, and have a wealth of data available that may be helpful for the general...
-
Outline the objectives and functions of memory management in operating systems. (Resource: Leverage Figures 7.1, 7.2, 7.3, and 7.4 from the textbook.) Compare and contrast the physical address...
-
Critically consider the internal and external ways in which a healthcare business can raise finance if it has a new product available with a Net Present Value of 100m Bahraini Dinar
-
Outline the purpose of an operating system (OS). Explain the interaction that takes place between an OS, applications, and computer hardware.?
-
You know that assets 1 and 2 have the same standard deviation and that the correlation between their returns is 0.4. You observe that the portfolio with 30% invested in asset 1 and the rest in asset...
-
Gilead Construction Inc., which is headquartered in Calgary, Alberta, built a small apartment building in Red Deer. The construction foreman, whose name was Jon Machenko, moved to Red Deer in May to...
-
An environmentalist wants to determine if the median amount of potassium (mg/L) in rainwater in Lincoln County, Nebraska, is different from that in the rainwater in Clarendon County, South Carolina....
-
A 1.509-g sample of a Pb/Cd alloy was dissolved in acid and diluted to exactly 250.0 mL in a volumetric flask. A 50.00-mL aliquot of the diluted solution was brought to a pH of 10.0 with a NH4+/NH3...
-
Calamine, which is used for relief of skin irritations, is a mixture of zinc and iron oxides. A 1.056-g sample of dried calamine was dissolved in acid and diluted to 250.0 mL. Potassium fluoride was...
-
A 3.650-g sample containing bromate and bromide was dissolved in sufficient water to give 250.0 mL. After acidification, silver nitrate was introduced to a 25.00-mL aliquot to precipitate AgBr, which...
-
In an engine, a piston oscillates with simple harmonic motion so that its position varies according to the expression x ( t ) = 15.0 cos( 20.0 t ) where x is in centimeters and t is in seconds. (a)...
-
When you think of your existence in the world, how do you think of it? How do you exist in the world? What is the world? What are you? What is the relationship between you and the world? Now consider...
-
Requirement 2 Drop Down Options: 1. Generating ORUsing 2.Divesting itself of ORInvesting in new 3. Borrowing OR Issuing Stock.......Borrowing OR Issuing 4. Decreased OR Increased 5. Strong OR Weak...

Study smarter with the SolutionInn App


