Titration of 50.00 mL of 0.04715 M Na 2 C 2 O 4 required 39.25 mL of
Question:
Titration of 50.00 mL of 0.04715 M Na2C2O4 required 39.25 mL of a potassium permanganate solution.
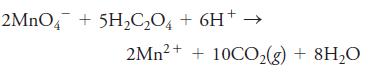
Calculate the molar concentration of the KMnO4 solution.
Transcribed Image Text:
2MNO4 + 5H2C,O, + 6H+ 2Mn?+ + 10CO,(g) + 8H20
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 28% (7 reviews)
For this redox titration we use the formu...View the full answer

Answered By

Marvine Ekina
Marvine Ekina
Dedicated and experienced Academic Tutor with a proven track record for helping students to improve their academic performance. Adept at evaluating students and creating learning plans based on their strengths and weaknesses. Bringing forth a devotion to education and helping others to achieve their academic and life goals.
PERSONAL INFORMATION
Address: , ,
Nationality:
Driving License:
Hobbies: reading
SKILLS
????? Problem Solving Skills
????? Predictive Modeling
????? Customer Service Skills
????? Creative Problem Solving Skills
????? Strong Analytical Skills
????? Project Management Skills
????? Multitasking Skills
????? Leadership Skills
????? Curriculum Development
????? Excellent Communication Skills
????? SAT Prep
????? Knowledge of Educational Philosophies
????? Informal and Formal Assessments
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Analytical Chemistry
ISBN: 9780357450390
10th Edition
Authors: Douglas A. Skoog, Donald M. West, F. James Holler, Stanley R. Crouch
Question Posted:
Students also viewed these Sciences questions
-
Calculate the molar concentration of a solution that is 50.0% NaOH (w/w) and has a specific gravity of 1.52.
-
Calculate the molar concentration of a 20.0% solution (w/w) of KCl that has a specific gravity of 1.13
-
Calculate the molar concentration of a dilute Ba(OH)2 solution if (a) 50.00 mL yielded 0.1791 g of BaSO4. (b) Titration of 0.4512 g of primary standard potassium hydrogen phthalate (KHP) required...
-
The pilot of a small boat charts a course such that the boat will always be equidistant from an upcoming rock and the shoreline. Describe the path of the boat. If the rock is 2 miles offshore, write...
-
Using the bond yield given in the final column of Table 15.2, verify the September 22, 2009, quoted price for the Province of New Brunswick 4.4% coupon bond, maturing June 3, 2019.
-
A genetic model describing inbreeding, in which mating takes place only between individuals of the same genotype, is given by the Markov process u(n+1) = T u(n), where is the transition matrix and...
-
Identify some of the critical motives and methods for the European expansion of the 16th century.
-
The records of Diamondale Company indicate a May 31 cash balance of $10,550, which includes undeposited receipts for May 30 and 31. The cash balance on the bank statement as of May 31 is $8,575. This...
-
Assume that you are the portfolio manager of the TTY Fund, a $5 million hedge fund that contains the following stocks. The required rate of return on the market is 12.00% and the risk-free rate is...
-
Use the sixth row of Table 1 in Appendix B to generate 12 random numbers between 1 and 99. Table 1 in Appendix B 92630 78240 9267 95457 53497 23894 37708 79862 79445 78735 71549 44843 26104...
-
The phosphorus in a 0.3019-g sample was precipitated as the slightly soluble (NH 4 ) 3 PO 4 . 12MoO 3 . This precipitate was filtered, washed, and then redissolved in acid. Treatment of the resulting...
-
A 0.7891-g sample of a mixture consisting solely of sodium bromide and potassium bromide yields 1.2895 g of silver bromide. What are the percentages of the two salts in the sample?
-
Suppose that the design specifications for hydraulic cylinder are 10.00 0.10 centimeters, and that the Taguchi loss function is estimated to be L(x) = 1,400 (x - T) 2 . (a) Determine the estimated...
-
Why did the sterling-based gold standard originally unravel?
-
Hope to release this to the market next year. The audit team has assessed inherent risk as Low, and control risk as Medium. What should Detection Risk be?
-
S wants to place property in an irrevocable trust for his 14-year-old granddaughter. The trust instrument provides for the accumulation of the trust income until the granddaughter reaches age 21, at...
-
What can be done to prevent financial abuse in older adults?
-
What is the major advantage of the kanban system?
-
Duke and Jacquie Coleman, married filing jointly, will establish a manufacturing business. The couple anticipates that the business will be profitable immediately due to a patent that Jacquie holds;...
-
Making use of the tables of atomic masses, find the velocity with which the products of the reaction B10 (n, ) Li7 come apart; the reaction proceeds via interaction of very slow neutrons with...
-
Indicate whether an aqueous solution of the following compounds is acidic, neutral, or basic. Explain your answer. *(a) NH4OAc (b) NaNO2 *(c) NaNO3 (d) NaHC2O4 *(e) Na2C2O4 (f) Na2HPO4 *(g) NaH2PO4...
-
Suggest an indicator that could be used to provide an end point for the titration of the first proton in H3AsO4.
-
Suggest a method for determining the amounts of H3PO4 and NaH2PO4 in an aqueous solution.
-
Oak Creek Furniture Factory (OCFF), a custom furniture manufacturer, uses job order costing to track the cost of each customer order. On March 1, OCFF had two jobs in process with the following...
-
Image transcription text 9:10 X Test_Belgeleme_Vak... Q E .. . Yapisal Test ve Validasyon Takimi Test Belgeleme Muhendisligi Aday Vaka Calismasi Ad: Soyad: 1) Asagidaki sekilde, kok noktasindan...
-
Individual Key Learning Report Individual Key Learning Report A+ Individual Report on Key Learnings Students are required to prepare an individual report at the end of the semester to present the...

Study smarter with the SolutionInn App


