Describe the reactions that convert epinephrine to the compound shown here. CH3 O OH 0
Question:
Describe the reactions that convert epinephrine to the compound shown here.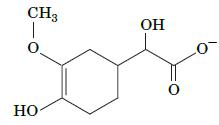
Transcribed Image Text:
CH3 O OH 0
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
The compound shown is vanillylmandelic acid VMA which is a metabolite of epinephrine Epinephrine is ...View the full answer

Answered By

Sarah Khan
My core expertise are:
-_ Finance
-_ Business
-_ Management
-_ Marketing Management
-_ Financial Management
-_ Corporate Finance
-_ HRM etc...
I have 7+ years of experience as an online tutor. I have hands-on experience in handling:
-_ Academic Papers
-_ Research Paper
-_ Dissertation Paper
-_ Case study analysis
-_ Research Proposals
-_ Business Plan
-_ Complexed financial calculations in excel
-_ Home Work Assistance
-_ PPT
-_ Thesis Paper
-_ Capstone Papers
-_ Essay Writing etc...
5.00+
91+ Reviews
92+ Question Solved
Related Book For 

Fundamentals Of Biochemistry Life At The Molecular Level
ISBN: 9781118918401
5th Edition
Authors: Donald Voet, Judith G Voet, Charlotte W Pratt
Question Posted:
Students also viewed these Sciences questions
-
Describe the reactions that are used in the steam-reforming process for the production of hydrogen.
-
THIRD AVENUE SOFTWARE HEALTH-CARE APP PROJECT This case is new for the ninth edition of Information Technology Project Management . The case provides an opportunity to apply agile and Scrum...
-
Epinephrine (adrenalin; see also Chapter 6 Opening) is produced in your body in a two-step process that accomplishes the transfer of a methyl group from methionine (Problem 70) to norepinephrine (see...
-
$ 45.00 Direct material: 5 pounds at $9.00 per pound Direct labor: 3 hours at $14 per hour Variable overhead: 3 hours at $9 per hour Total standard variable cost per unit 42.00 27.00 $114.00 The...
-
Why is the server segment of the computer industry becoming so competitive today? SGI, or Silicon Graphics International, was formed by the merger of Rackable Systems and Silicon Graphics in May...
-
Richardson Services Ltd. was formed on May 1, 2017. The following transactions took place during the first month. Transactions on May 1: 1. Shareholders invested 50,000 cash in exchange for ordinary...
-
The interfacial shear stresses, \(\tau\), and the fiber normal stresses, \(\sigma_{f}\) acting on a differential element at a distance \(x\) from the end of the fiber are shown in Figure 6.4, where...
-
Refer to the data in Exercise 6-31. The president of Tiger Furnishings is confused about the differences in costs that result from using direct labor costs and machine-hours. Required a. Explain why...
-
A boat (with a flat bottom) and its cargo weigh 5,200 N. The area of the boat's bottom is 8 m 2 . How far below the surface of the water is the boat's bottom when it is floating in water? Answer in...
-
From which amino acid is 2-phenylethanol derived and what chemical changes take place in the conversion? OH 2-Phenylethanol
-
Many of the most widely used herbicides inhibit the synthesis of aromatic amino acids. Explain why the compounds are safe to use near animals.
-
In 1996, Marriott International made an issue of unusual bonds called liquid yield option notes, or LYONS. The bond matured in 2011, had a zero coupon, and was issued at $532.15. It could have been...
-
Find the Time Complexity of the following Algorithms. For (i=0; i*i
-
Gravel is being dumped from a conveyor belt at a rate of 25 ft/min, and its coarseness is such that it forms a pile in the shape of a cone whose base diameter and height are always equal. How fast is...
-
Last year, a battery manufacturing company in Toronto incurred a loss of $755,000.00 by producing and selling 42,000 batteries. If their total revenue for the year was $17,000,000.00 and the...
-
Task 1 Task 2 a = 1, when the exponent on a increases, the power itself increases, provided a is greater than 1. It follows that if the exponent is infinitely small and positive, then the power also...
-
Given an array of 20 elements 10, 21, 0, 41, 24, 9, 29, 0, 90, 85, 77, 56, 62, 17, 0, 33, 23, 44, 50, 52. Complete the program to print the total number of odd numbers in the array and even numbers...
-
Heineken N.V. which is a global brewer domiciled in the Netherlands, reports the following balance sheet accounts for the year ended December 31, 2013 (euro in millions). Prepare the balance sheet...
-
As you rewrite these sentences, replace the cliches and buzzwords with plain language (if you don't recognize any of these terms, you can find definitions online): a. Being a jack-of-all-trades, Dave...
-
Muscalure is the sex pheromone of the common housefly and has the molecular formula C 23 H 46 . When treated with O 3 followed by DMS, the following two compounds are produced. Draw two possible...
-
Propose a plausible mechanism for each of the following reactions: a. b. stitl. [H,SO,] Conc. H2SO4
-
Suggest an efficient synthesis for the following transformation: CI H
-
Solve ADEF. Round to the nearest whole number when necessary. Give angle measures in degrees. 4 E |DE= 4 mZDz DF mZE= 90 EF= 6 m/F 9 LL F
-
Find the total energy available from a rechargeable 1.25-V flashlight battery with a 1.2-Ah rating. If all the energy in a 9-V transistor radio battery rated at 0.392 Ah is used to lift a 150-lb man,...
-
The organization that Mike works in finds that one of their domains is directing traffic to a competitors website. When Mike checks, the domain information has been changed, including the contact...

Study smarter with the SolutionInn App


