The reversible reaction shown here is part of the Calvin cycle, a pathway in photosynthetic organisms. Which
Question:
The reversible reaction shown here is part of the Calvin cycle, a pathway in photosynthetic organisms. Which glycolytic reaction does this reaction resemble and what type of enzyme catalyzes it?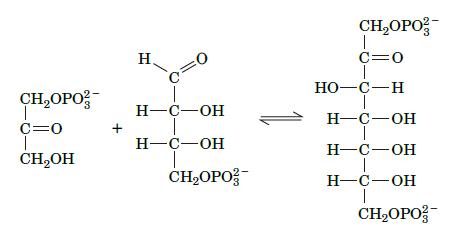
Transcribed Image Text:
CHOPO 1 C=0 CHOH + H =0 H-C-OH H-C-OH CHOPO CHOPO T C=0 HO-C-H H-C-OH H-C-OH H-C-OH T CHOPO
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
The reversible reaction shown in the Calvin cycle that resembles a glycolytic reaction is t...View the full answer

Answered By

BRIAN MUSINGA
I possess a Bachelors of Commerce degree(Marketing option) and am currently undertaking an MBA in marketing. I believe that I possess the required knowledge and skills to tutor in the subject named. I have also written numerous research academic papers much to the satisfaction of clients and my professors.
5.00+
2+ Reviews
17+ Question Solved
Related Book For 

Fundamentals Of Biochemistry Life At The Molecular Level
ISBN: 9781118918401
5th Edition
Authors: Donald Voet, Judith G Voet, Charlotte W Pratt
Question Posted:
Students also viewed these Sciences questions
-
Step 4 of the pentose phosphate pathway converts ribulose-5- hosphate to ribose-5-phosphate. Which glycolytic reaction does this reaction resemble and what type of enzyme catalyzes it?
-
The bacterial enzyme polynucleotide phosphorylase (PNPase) is a 3 S exoribonuclease that degrades mRNA. (a) The enzyme catalyzes a phosphorolysis reaction, as does glycogen phosphorylase (Section...
-
The Earth has been getting warmer. Most climate scientists agree that one important cause of the warming is the increase in atmospheric levels of carbon dioxide (CO2), a greenhouse gas. Here is part...
-
Identify possible opportunities for denormalizing these relations as part of the physical design of the database. Which ones would you be most likely to implement
-
The capital investment committee of Eastern Trucking Inc. is considering two investment projects. The estimated income from operations and net cash flows from each investment are as follows: Each...
-
A tall standpipe with an open top (see figure) has diameter d = 2.2 m and wall thickness t = 20 mm. (a) What height h of water will produce a circumferential stress of 12 MPa in the wall of the...
-
Females Rating Males on OKCupid The OKCupid dating site provides lots of very interesting data. Figure 2.61 shows a scatterplot of the age of males that females find most attractive, based on the age...
-
Blackerby Photo reported the following figures on its December 31, 2014, income statement and balance sheet: Compute the asset turnover ratio for2014. Net sales 462,000 Dec. 31, 2014 $ 15,000 54,000...
-
Evaluate using integration by parts: f^ (x. In (5x))dx. (x In (5.x)) dx = Click here to start next step (x-In (5-x))dx = +
-
In a mixture of NAD+, NADH, ubiquinone, and ubiquinol, which compound will be oxidized? Which will be reduced?
-
The thioester described in Problem 29 reacts readily with compounds with the formula ROH or RNH2. Draw the resulting ester and amide reaction products. Problem 29 Some proteins contain internal...
-
Stacey Ellis is paid at the following differential piece rate: 1-100, $2.58; 101-250, $2.72; 251 and up, $3.15. Find her gross earnings for completing 475 pieces.
-
A ball is pressed down on a spring and then released from rest. The spring launches the ball upward. Identify the energy conversions that occur between the instant the ball is released and the...
-
A uniform chain of inertia \(m\) and length \(\ell\) is lying on a slippery table. When one quarter of its length hangs over the edge, the chain begins to slip off. How fast is it moving when the...
-
Is it possible to figure out an object's past motion if you know its present position and velocity and the history of all the forces exerted on it?
-
If a single known force is exerted on an object of unknown initial velocity for a certain time interval, you know the change in the object's momentum. However, you don't know its change in kinetic...
-
(a) When you stretch a Slinky spring a moderate amount and then release it, it returns to its original state, and all the potential energy stored by the stretching is converted to another form. If...
-
Discuss legal and scope parameters that might impact a computer forensics engagement.
-
MgO prevents premature evaporation of Al in a furnace by maintaining the aluminum as Al2O3. Another type of matrix modifier prevents loss of signal from the atom X that readily forms the molecular...
-
The reaction catalyzed by malate dehydrogenase, has a G°' = 4.6 kJ mol-1. (a) Would this reaction occur spontaneously in a cell? (b) How does the citrate synthase reaction promote the malate...
-
Predict whether creatine kinase will operate in the direction of ATP synthesis or phosphocreatine synthesis at 25C when [ATP] = 4 mM, [ADP] = 0.15 mM, [phosphocreatine] = 2.5 mM, and [creatine] = 1...
-
If intracellular [ATP] = 5 mM, [ADP] = 0.5 mM, and [Pi] = 1.0 mM, calculate the concentration of AMP at pH 7 and 25C under the condition that the adenylate kinase reaction is at equilibrium.
-
Atari needs $4.5 million in new investment capital to develop and market downloadable game software for its new GPS2-ZX system. The plan is to sell $10,000 face-value corporate bonds at a discount of...
-
Six months ago, you purchased a new pickup truck. At the time, you were a little light on cash and decided to finance the purchase. You financed a total of $24,500 with a 60-month amortized loan...
-
What are the molecular pathways involved in immunological tolerance, including central and peripheral mechanisms that prevent self-reactive lymphocytes from causing autoimmune reactions, and how do...

Study smarter with the SolutionInn App


