Which fatty acids were used to build the molecule shown here, which functions as a signaling molecule?
Question:
Which fatty acids were used to build the molecule shown here, which functions as a signaling molecule?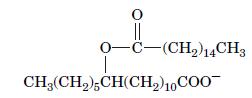
Transcribed Image Text:
0-C-(CH) 14CH3 CH(CH),CH(CH)10 COO
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 83% (6 reviews)
The fatty acid used to build the molecule shown in the image is arachidonic acid an omega6 fatty aci...View the full answer

Answered By

S Mwaura
A quality-driven writer with special technical skills and vast experience in various disciplines. A plagiarism-free paper and impeccable quality content are what I deliver. Timely delivery and originality are guaranteed. Kindly allow me to do any work for you and I guarantee you an A-worthy paper.
4.80+
27+ Reviews
73+ Question Solved
Related Book For 

Fundamentals Of Biochemistry Life At The Molecular Level
ISBN: 9781118918401
5th Edition
Authors: Donald Voet, Judith G Voet, Charlotte W Pratt
Question Posted:
Students also viewed these Sciences questions
-
Which fatty acids were used to build the molecule shown here, which is synthesized by intestinal cells following a meal? ZH N O
-
The molecule shown here is difluoromethane (CH2F2), which is used as a refrigerant called R-32. (a) Based on the structure, how many electron domains surround the C atom in this molecule? (b) Would...
-
CK investments (Pty) is a local hand sanitizing production company. The company was incorporated in 2010. The company was started by Charles Mujende who is the 100% shareholder. CK is run by the...
-
Steven Stores, Inc. provided the following statement of net income for the current year. All income is subject to a 40% income tax rate. The company also had $ 735 of unrealized holding gains on its...
-
Under what conditions might concentration on a single business be inconsistent with the goal?
-
An inexperienced accountant for Zoeng Company, Ltd. made the following errors in recording merchandising transactions. 1. A HK$1,750 refund to a customer for faulty merchandise was debited to Sales...
-
Describe an experiment, and give the necessary equations for the measurement of the complex longitudinal modulus, \(E_{\mathrm{f} 1}^{*}\), of a reinforcing fiber.
-
Methane reacts with chlorine to produce methyl chloride and hydrogen chloride. Once formed, the methyl chloride may undergo further chlorination to form methylenc chloride (CH 2 CI 2 ), chloroform,...
-
Conduct investigation into current trends in healthcare using the BU Library search capabilities and any other materials/resources provided to you in the course. Discuss what you think are some of...
-
Why are the symptoms of carnitine palmitoyl transferase II deficiency more severe during fasting?
-
Two lipoproteins have the following characteristics: Which lipoprotein has a higher density? Lipoprotein A Lipoprotein B Diameter () 200 100 % Triacylglycerol 10 5 % Protein 20 55
-
In passive solar heating, the goal is to absorb heat from the sun during the day and release it during the night. Which material would be better for this application: one with a high heat capacity or...
-
Davis Inc. has fixed operating costs of $1,000,000 and variable costs of $80 per unit. If it sells the product for $100 per unity, what is the breakeven quantity? State Flag Co.has a beta of 1.40....
-
The torque (or moment) caused by a force F = (256 +325) N acting at position r = (-1.552 - 1.55 ) m relative to the axis of rotation (pivot) is 7 = F x F. Find the torque caused by F acting at...
-
Indigo Depot is having a clearance sale of their fall items. All clearance items are marked an additional 30 % off the sale price. A jacket that originally costs $ 140 before any discount is on sale...
-
What is the current total product cost (for the 20 million units) including fixed and variable cost? Great-Cola spends $3 on direct materials, direct labor, and variable manufacturing overhead for...
-
Determine the moment of force of240 lb regarding: a) To point A (climb) b) At the linnea DB(vectorial)
-
Swiss Group reports net income of $40,000 for 2015. At the beginning of 2015, Swiss Group had $200,000 in assets. By the end of 2015, assets had grown to $300,000. What is Swiss Groups 2015 return on...
-
Complete the following acid-base reactions: (a) HCCH + NaH
-
Predict the final product(s) for each sequence of reactions: (a) (b) (c) (d) Br Br. 1) Excess NaNH, 2) EECI 3) H2, Lindlar's catalyst 1) NANH, 2) Mel 3) 9-BBN 4) H202, NaOH H-CEC-H
-
When (R)-4-bromohept-2-yne is treated with H 2 in the presence of Pt, the product is optically inactive. Yet, when (R)-4-bromohex-2-yne is treated with the same conditions, the product is optically...
-
If the reaction Fe 2 N(s) + 3/2H 2 (g) 2Fe(s) + NH 3 (g) comes to equilibrium at a total pressure of 1 bar, analysis of the gas shows that at 700. and 800. K, PNH 3 /PH 2 = 2.165 and 1.083,...
-
Vinamilk has the following information at the end of the year 2019: Total dividends $21,460; beta 0.91; 6,500 shares outstanding; current market price is $50. Assume that the risk free rate is 5%,...
-
The capital budgeting process is comprehensive and is based on certain assumptions, models, and benchmarks. This process often begins with a project analysis. Generally, the first step in a capital...
-
Give the definition of the marginal rate of substitution. b. Give the definition of indifference curve. c. Hai Ying loves chestnuts but despises strawberries - chestnuts are a "good" for her and...

Study smarter with the SolutionInn App


