Which type of enzyme catalyzes the following reactions? (a) COO (b) CONADH + H+ CH3 COO COO
Question:
Which type of enzyme catalyzes the following reactions?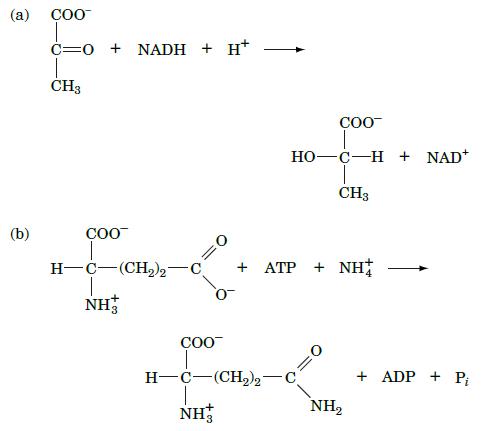
Transcribed Image Text:
(a) COO (b) CONADH + H+ CH3 COO COO HO-C-H + NAD* COO H-C(CH)2 C + ATP + NH+ NH H-C-(CH)2 C NH3 CH3 NH + ADP + Pi
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
a COO CO NADH H CH3 COO HOCH NAD CH3 COO The reaction youve shown appears to ...View the full answer

Answered By

Muhammad Ghyas Asif
It is my obligation to present efficient services to my clients by providing a work of quality, unique, competent and relevant. I hope you have confidence in me and assign me the order and i promise to follow all the instructions and keep time.
4.60+
109+ Reviews
203+ Question Solved
Related Book For 

Fundamentals Of Biochemistry Life At The Molecular Level
ISBN: 9781118918401
5th Edition
Authors: Donald Voet, Judith G Voet, Charlotte W Pratt
Question Posted:
Students also viewed these Sciences questions
-
Which type of enzyme catalyzes the following reactions? (a) COO H-C-CH3 NH (b) COO- T C=O + H+ CH3 HC-C-H NH H COO C=0+0=C=0 1 CH3
-
a. List two common electron carriers used in biological cells and summarize the role they play in the production of ATP. b. Based upon the information in table, explain which type or types of enzyme...
-
The role of Joseph Ripp in the accounting fraud at AOL is one of whether a CFO who seemingly goes along with an accounting fraud and then is responsible for uncovering it should be viewed as a hero...
-
Find all conjugates in C of the given number over the given field. 1+2 over Q
-
a. Explain the advantages of using GAS to assist with audits and give several examples of how it may be used. b. Describe the audit purpose facilitated and the procedural steps to be followed when...
-
(a) After the label fell off a bottle containing a clear liquid believed to be benzene, a chemist measured the density of the liquid to verify its identity. A 25.0-mL portion of the liquid had a mass...
-
In Example 3.1, we see that \(27.5 \%\) of US adults are college graduates. (a) Use StatKey or other technology to generate a sampling distribution for the sample proportion of college graduates...
-
Stockman Company began 2010 with three jobs in progress: During 2010, the following transactions occurred: a. The firm purchased and paid for $542,000 of raw material. b. Factory payroll records...
-
You have an outstanding student loan with required payments of $550 per month for the next four years. The interest rate on the loan is 11% APR (compounded monthly). Now that you realize your best...
-
Approximately how much does staphylococcal nuclease decrease the activation free energy G of its reaction (the hydrolysis of a phosphodiester bond) at 25C?
-
Choose the best description of an enzyme: (a) It allows a chemical reaction to proceed extremely fast. (b) It increases the rate at which a chemical reaction approaches equilibrium relative to its...
-
Find the average value of the following functions over the given range. Sketch a graph of the function along with a horizontal line at the average to make sure that your answer makes sense. 1/x for...
-
Let us consider a European-style put option on a non-dividend-paying asset whose price follows a GBM with drift 10% and volatility 40% (annualized). The risk-free rate is 5% with continuous...
-
Why do you think the atria of the heart are less muscular than the ventricles? Why is the left ventricle more muscular than the right ventricle?
-
Explain what happens when a B cell first encounters a pathogen and binds to an antigen on the pathogen.
-
How do you inhale?
-
How does fluid move from the circulatory system into a nephron?
-
1. Which of the following is not a characteristic of a profession? a. It enjoys a high level of public trust and confidence. b. It is organized into associations. c. There are published authoritative...
-
What recommendations would you make to Big Four firms to help them (1) avoid confrontations with governmental officials in an authoritarian society and (2) deal effectively with such confrontations...
-
Purple photosynthetic bacteria have different pigments than higher plants. Why is this an advantage for these bacteria?
-
What distinguishes the chlorophyll in a reaction center from the antennae chlorophyll?
-
The initial electron transfers in the bacterial photosynthetic reaction center are extremely rapid, but the lifetime of the terminal semiquinone is relatively long. (a) Why is it essential for the...
-
A horse requires 5 mEq of potassium gluconate two times daily for 28 days. Each tablet contains 10 mEq of potassium gluconate. Show all steps and include units for full credit. Round your answers to...
-
Answer For the series diode configuration in the figure below, determine VD, VR and ID. [Total Marks 2] Si R2.2 k Note: Since the applied voltage establishes a current in the clockwise direction...
-
Sebastian is in France for his vacation. He would like to buy a beautiful leather jacket for 500 Euro. The exchange rate is $1 USD = .81 Euro. Calculate the price in USD.

Study smarter with the SolutionInn App


