A mixture that contains 40% by mole n-heptane in n-decane is to be separated in a series
Question:
A mixture that contains 40% by mole n-heptane in n-decane is to be separated in a series of flush separators until a stream is obtained that contains at least 95% n-heptane. Determine the number of separators needed, their temperature, and the recovery of n-heptane if all separators are at 1.013 bar and V/L = 3 in all separators. Txy data are given below:
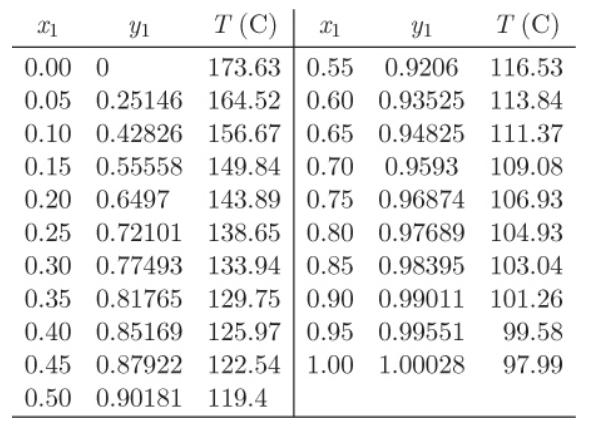
Transcribed Image Text:
X1 T (C) X1 Yı T (C) 0.00 0 173.63 0.55 0.9206 116.53 0.05 0.25146 164.52 0.60 0.93525 113.84 0.10 0.42826 156.67 0.65 0.94825 111.37 0.15 0.55558 149.84 0.70 0.9593 109.08 0.20 0.6497 143.89 0.75 0.96874 106.93 0.25 0.72101 138.65 0.80 0.97689 104.93 0.30 0.77493 133.94 0.85 0.98395 0.35 0.81765 129.75 0.90 0.99011 0.40 0.85169 125.97 0.95 0.99551 0.45 0.87922 122.54 1.00 103.04 101.26 1.00028 0.50 0.90181 119.4 Y1 99.58 97.99
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
To determine the number of separators needed their temperature and the recovery of nheptane in each separator we can use the concept of flash distilla...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Chemical Engineering Thermodynamics
ISBN: 9780132693066
1st Edition
Authors: Themis Matsoukas
Question Posted:
Students also viewed these Engineering questions
-
A steady stream of equimolar N2 and CO2 mixture at 100 kPa and 18C is to be separated into N2 and CO2 gases at 100 kPa and 18C. Determine the minimum work required per unit mass of mixture to...
-
Journalize the 10 transactions for your company using excel (must include descriptions). Your opening entry should include the following 4 items: Bank $10,000 Supplies $5,000 Bank Loan $20,000...
-
A total of 2,000 gallons of 70 wt% ethanol in water, having a specific gravity of 0.871, is to be separated at 1 atm in a batch rectifier operating at constant distillate composition with a constant...
-
What is a buy-sell agreement, and how does life insurance facilitate it?
-
The government places a tax on the purchase of socks. a. Illustrate the effect of this tax on equilibrium price and quantity in the socks market. Identify the following areas both before and after...
-
Generate the first simplex iteration of Example 4.2-1 (you may use TORA's Iterations M-method for convenience), then use Formulas 1 and 2 to verify all the elements of the resulting tableau.
-
It seems that there are many ways for fraud and abuse to take place in the field of accounting. How can this occur when there are so many regulatory bodies and professional accounting standards in...
-
Presented below is the income statement of Cowan, Inc.: Sales ................$380,000 Cost of goods sold .......... 225,000 Gross profit .............$155,000 Operating expenses .......... 85,000...
-
At the beginning of each day, the Factory General Manager consults with the sales department and determines which jobs will be completed for the day. The General Manager forwards the Job Cost Sheet...
-
A stream that contains a mixture of methane (25% by mol) and carbon monoxide is compressed from 1 bar, 35 to 12 bar. The compressor efficiency is 90%. Treating the mixture as an ideal gas, calculate...
-
Use the data below for the system ethyl propyl ether (1)-chloroform (2) to answer the following questions: a) What is the boiling point of chloroform at 0.5 bar? b) Is this a maximum boiling or...
-
Suppose you use an average of 500 kW.h of electric energy per month in your home. (a) How long would 1.00 g of mass converted to electric energy with an efficiency of 38.0% last you? (b) How many...
-
In 2005, before the financial crisis, Timothy Geithner, who was then president of the Federal Reserve Bank of New York, thought that leverage at hedge funds was rising, probably because of heightened...
-
In late 2009, during the debate over the Dodd-Frank Act, a newspaper article noted: Last summer, the central bank hired an experienced Democratic hand and former lobbyist, Linda Robertson, to help...
-
An article in the Economist magazine says about investment banks: By unlocking the capital markets and helping firms to manage risks, investment banks are important conduits of credit. How do...
-
Are the high rates of inflation that the United States experienced during the 1970s consistent with the public interest view of the Feds motivation?
-
A bank analyst was quoted as saying: There are very real expenses to owning and operating ATMs. In the current environment, banks are unable to support the cost structure they have historically. a....
-
You can borrow $5,000 for 60 days with an interest payment of $125. What is the simple rate of interest? What is the compound rate of interest?
-
Kenneth Hubbard has prepared the following list of statements about managerial accounting and financial accounting. 1. Financial accounting focuses on providing information to internal users. 2....
-
Several processes are described. Indicate whether each is reasonably modeled as reversible, and if not, indicate what aspect of the process makes it irreversible. A . The inside of a refrigerator is...
-
A steady-state distillation column is designed to separate benzene from toluene. The separation is nearly enough complete that, for the purposes of designing the reboiler and the condenser, we can...
-
A liquid stream contains 1 lb m /s of the compound at T = 100F and P = 1 atm. It needs to be boiled and heated to P = 1 atm and 175F, as that is the temperature at which it must enter a chemical...
-
(i) Give ONE example of a constant-pressure process that is relevant to materials science. (ii) Give ONE example of a constant-volume process that is relevant to materials science. Explain the...
-
3. Reactant purification The CAT2 catalyst requires an undesirable impurity, S, in the feed of the packed bed reactor to be removed before entering the reactor. This is done by separating S with a...
-
2. CAT2 technology with non-porous catalyst You have invented a completely new catalyst for the CAT2 technology. This is based on solid (not porous) catalyst pellets. For this catalyst, the reaction...

Study smarter with the SolutionInn App


