An ideal gas has C P *=(7/2)R. One mole of this gas is confined in a piston-cylinder
Question:
An ideal gas has CP*=(7/2)R. One mole of this gas is confined in a piston-cylinder device. Initially, the gas is at T = 300 K and P = 1 bar (Figure 3-13). If the gas is compressed isothermally to P = 5 bar, find the amounts of work and heat associated with the process.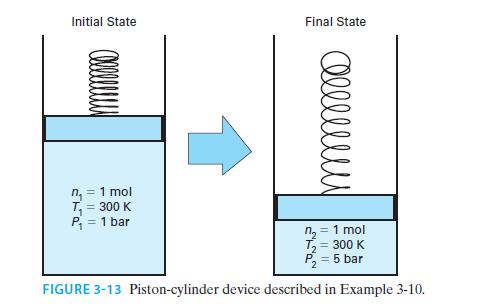
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
Step 1 Define the system Define the system as the gas inside the cylinder Step 2 Apply and simplify ...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

Fundamentals Of Chemical Engineering Thermodynamics
ISBN: 9781111580704
1st Edition
Authors: Kevin D. Dahm, Donald P. Visco
Question Posted:
Students also viewed these Engineering questions
-
A fundamental aspect of Six Sigma methodology is the identification of critical to quality (CTQ) characteristics that are vital to customer satisfaction. Identify and depict the various activities...
-
Refer to M3-5 for information regarding Stone Company. Using the FIFO method, reconcile the number of physical units and calculate the number of equivalentunits. Physical Units 70,000 92,000 Percent...
-
James came back from Overseas to seek full-time employment in Singapore. He has the following stock options which were granted to him in 2021 and 2022: In 2021, a stock option for 10,000 shares at an...
-
Finally, Reza mentions that he was hired into the CFO role on an interim basis with the possibility of being made permanent based on performance. Although he wants to succeed in this role, he does...
-
Consider Review Exercise 9.101. Let us assume that the data have not been collected yet. Let us also assume that previous statistics suggest that 1 = 2 = $4000. Are the sample sizes in Review...
-
Consider the nuclear reaction where X is a nuclide 21He + 94Be X + 42He (a) What are the values of Z and A for the nuclide X? (b) How much energy is liberated? (c) Estimate the threshold energy for...
-
P(54 < x < 64) Write the binomial probability in words. Then, use a continuity correction to convert the binomial probability to a normal distribution probability.
-
Rouse Company, a real estate developer, is well known as one of the few U.S. companies to have reported the current value of property and equipment in its financial statements. As mentioned in the...
-
Prior to beginning work on this discussion, please read about the Economic Value Added (EVA) and the Balanced Scorecard management systems on the websites of theCorporate Finance Institute (CFI) and...
-
The balloon portion of a dirigible contains 5000 moles of helium. Initially, the helium is at T = 15C and P = 0.95 atm, and is 500 m off the ground (where the atmospheric pressure is also 0.95 atm)....
-
A tank has a volume of 500 liters and is initially full of a liquid that is at 300 K, has a density of 0.8 kg/L, and a constant heat capacity of C V = 3 J/g K. The tank can be modeled as perfectly...
-
Describe the activities performed by the five modules of the SAP system.
-
How should the interfaces handle errors?
-
What should we do to ensure the system users are aware of what to do in the system?
-
What are the differences between sequence diagrams and collaboration diagrams?
-
What does it mean to test the system on actual users?
-
Why can some user interfaces cause users to feel confused, panicky, or frustrated?
-
In 2002, Mayola Williams, the widow of a man who smoked as many as 3 packs of Marlboro cigarettes a day for almost 50 years, sued Philip Morris USA for the torts of negligence and fraud. Williams's...
-
Show that every group G with identity e and such that x * x = e for all x G is abelian.
-
Represent each of the following combinations of unit in the correct SI form: (a) Mg/ms, (b) N/mm, (c) mN/(kgs).
-
Convert: (a) 200 lb ft to N.m (b) 350 lb/ft 3 to kN/m 3 (c) Express the result to three significant figures. Use an appropriate prefix.
-
Represent each of the following as a number between 0.1 and 1000 using an appropriate prefix: (a) 45320 kN, (b) 568(105) mm, (c) 0.00563 mg.
-
An online software store just had a sale. The percentage discount on their items was given based on the following chart: Price Discount $0-$15 5% >$15 and $35 The end-user wants to figure out how...
-
The Farmer's Market just paid an annual dividend of $5 on its stock. The growth rate in dividends is expected to be a constant 5 percent per year indefinitely. Investors require a 13 percent return...
-
To answer the following questions, perform an Internet search for sample proposals. For example, type project proposal example into Google or Yahoo. Based on the results of your search, find a sample...

Study smarter with the SolutionInn App


