Determine the temperature and phase of water from the following information. If the phase is a vapor-liquid
Question:
Determine the temperature and phase of water from the following information. If the phase is a vapor-liquid mixture, report the fraction of vapor and liquid.
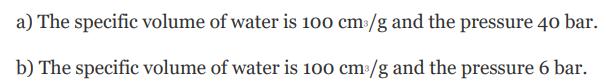
Transcribed Image Text:
a) The specific volume of water is 100 cm /g and the pressure 40 bar. b) The specific volume of water is 100 cm/g and the pressure 6 bar.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
To determine the temperature and phase of water given the specific volume and pressure we can use th...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Chemical Engineering Thermodynamics
ISBN: 9780132693066
1st Edition
Authors: Themis Matsoukas
Question Posted:
Students also viewed these Engineering questions
-
As shown in Figure, water (W) and n-butanol (B) can form a three-phase system at 101 kPa. For a mixture of overall composition of 60 mol% W and 40 mol% B, use a simulation computer program and the...
-
A liquid mixture containing 70.0 mol of n -pentane and 30.0 mol of n -hexane initially at 46?C is partially vaporized at P = 1 atm in a single-stage distillation apparatus (Rayleigh still). The heat...
-
Water at 50F and 50 psia is heated in a chamber by mixing it with saturated water vapor at 50 psia. If both streams enter the mixing chamber at the same mass flow rate, determine the temperature and...
-
(7b-4c-1)-2 (2b3c-2)5 Simplify.
-
CGK Rheosystems makes high-performance rotational viscometers capable of steady shear and yield stress testing in a rugged, compact footprint. How much could the company afford to spend now on new...
-
Suppose you have surveyed 30 people and asked them whether they are white (W) or nonwhite (N), and how many traumas (serious accidents, rapes, or crimes) they have experienced in the past year. You...
-
Review the list of common closing mistakes in the chapter. Give specific examples of how each might affect your success in a sales call.
-
1. What other management development approaches could the firm use to improve managerial skills other than I-MBA programs? 2. What criteria should the firm use to determine if it will pay for...
-
Provide in detail the process for contesting OSHA citations.
-
The following data are available for a gas: To calculate the molar volume at 25 C, 12 bar, our team came up with two different suggestions: (a) Linear interpolation for Vbetween the given pressure, ...
-
In 1656, Otto von Guericke of Magdeburg presented his invention, a vacuum pump, through a demonstration that became a popular sensation. A metal sphere made of two hemispheres (now known as the...
-
A school district forbids students from using their cell phones on school buses, but many students ignore this rule. A frustrated bus driver installs a cell phone jammer on his bus. When the jammer...
-
Suppose you are Samuel Cook, CEO of DeFlava Coffee. DeFlava currently operates in south-eastern Australia, and you are considering opening a factory and sales office in Queensland. What questions do...
-
Thomas received $30,000 in a legal settlement in 2017. The tax treatment of the item is not certain. Thomass research results were ambiguous and he is not sure if the income is taxable. Because some...
-
You are the CEO of a Japanese importer that resells designer bags, purses, and wallets across Southeast Asia. Many of the products are manufactured in Southeast Asia, mainly Vietnam, Indonesia, and...
-
Suppose your brother, the owner of Muscle Up, a fitness centre, has asked you for a substantial loan to help him expand his business. Required: What would you like to know about Muscle Up before you...
-
Cynthia and Howard, married taxpayers filing a joint return, have $100,000 in taxable income in 2017. They have 4 children (ages 4 through 12) who have no income that is taxable. If they can legally...
-
What does the term qualified indicate when describing a pension plan? Why is it important that a pension plan be qualified?
-
What is the difference between adsorption and absorption?
-
A two-component, two-phase mixture has how many degrees of freedom, according to Gibbs phase rule?
-
What is the entropy change when you mix 1 mole of 1-propanol with 4 moles of ethanol at 25C and 1 atm? Assume an ideal solution.
-
Using the data for the benzene (1) + 2-propanol (2) system in Table 9-3, fit the experimental data to an appropriate polynomial equation whose independent variable is the mole fraction of ethanol....
-
As recently as 20,000 years ago a continental glacier extended as far south as the central United States. Describe what impacts the ice might have had on the average position and magnitude of the...
-
Option Expiration Go to www.cboe.com, highlight the Trading Tools tab, then follow the Expiration Calendar link. On what day do equity options expire in the current month? On what day do they expire...
-
Kurt will receive $1,200 a month for five years from an insurance settlement. The first payment was received today. If he invests the full amount of each payment at a guaranteed 6.15 percent rate,...

Study smarter with the SolutionInn App


