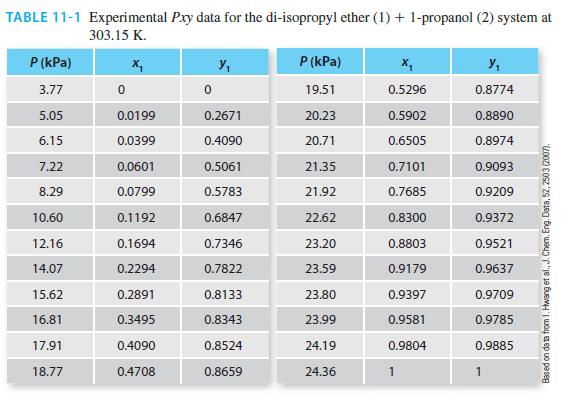
Perform a reduction of the data in Table 11-1 for the di-isopropyl ether (1) + 1-propanol (2)
Question:
Perform a reduction of the data in Table 11-1 for the di-isopropyl ether (1) + 1-propanol (2) system at 303.15 K using both the 1-parameter and 2-parameter Margules equations.
Transcribed Image Text:
TABLE 11-1 Experimental Pxy data for the di-isopropyl ether (1) + 1-propanol (2) system at 303.15 K. X₂ P (kPa) 3.77 5.05 6.15 7.22 8.29 10.60 12.16 14.07 15.62 16.81 17.91 18.77 0 0.0199 0.0399 0.0601 0.0799 0.1192 0.1694 0.2294 0.2891 0.3495 0.4090 0.4708 0 Y₁ 0.2671 0.4090 0.5061 0.5783 0.6847 0.7346 0.7822 0.8133 0.8343 0.8524 0.8659 P (kPa) 19.51 20.23 20.71 21.35 21.92 22.62 23.20 23.59 23.80 23.99 24.19 24.36 0.5296 0.5902 0.6505 0.7101 0.7685 0.8300 0.8803 0.9179 0.9397 0.9581 0.9804 y₁ 0.8774 0.8890 0.8974 0.9093 0.9209 0.9372 0.9521 0.9637 0.9709 0.9785 0.9885 1 Based on data from 1. Hwang et al., J. Chem. Eng. Data, 52, 2503 (2007).
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Step 1 Obtain the experimental activity coefficients and the experimental excess molar Gibbs free energy We can obtain the experimental activity coefficients and the excess molar Gibbs free energy in ...View the full answer

Answered By

William Otieno
I am a professional tutor and a writer with excellent skills that are important in serving the bloggers and other specialties that requires a great writer. The important aspects of being the best are that I have served so many clients with excellence
With excellent skills, I have acquired very many recommendations which have made it possible for me to survive as an excellent and cherished writer. Being an excellent content writer am also a reputable IT writer with essential skills that can make one turn papers into excellent result.
4.70+
83+ Reviews
354+ Question Solved
Related Book For 

Fundamentals Of Chemical Engineering Thermodynamics
ISBN: 9781111580704
1st Edition
Authors: Kevin D. Dahm, Donald P. Visco
Question Posted:
Students also viewed these Engineering questions
-
The article Seismic Hazard in Greece Based on Different Strong Ground Motion Parameters (S. Koutrakis, G. Karakaisis, et al., Journal of Earthquake Engineering, 2002:75109) presents a study of...
-
Using the data in Table 116 on page 300, indicate the semiannual interest payment dates for the Motorola bonds that mature in 2031. (For the item in question, look under Interest Dates.) The two...
-
The data in Table P-13 are closing stock quotations for the DEF Corporation for 150 days. Determine the appropriate ARIMA model, and forecast the stock price five days ahead from forecast origin t =...
-
Assume you have just been hired as a business manager of PizzaPalace, a regional pizza restaurant chain. The companys EBIT was $120 million last year and is not expected to grow. PizzaPalace is in...
-
Why is it important for a project to have flexibility?
-
Regal Rooms Ltd is a rug and drapery retailer. Regal Rooms specialises in selling floor rugs and window dressings. The following information was derived from the shops accounting records for the year...
-
The following reactions take place at \(1500 \mathrm{~K}\) and \(10 \mathrm{bar}\) : \[ \begin{align*} & A+B ightarrow C+D \tag{A}\\ & A+C ightarrow 2 E \tag{B} \end{align*} \] where...
-
Tony Masasi started his own consulting firm, Masasi Company, on June 1, 2010. The trial balance at June 30 is shown below. In addition to those accounts listed on the trial balance, the chart of...
-
A company can pay the fire insurance premiums on its factory either quarterly or annually. If paid annually, the premium is $3600, paid at the beginning of the year. If paid quarterly, there is a $30...
-
Estimate the vapor-liquid equilibrium for the n-pentane (1) + benzene (2) system at 313.15 K using the van Laar equation.
-
Estimate the mole fraction of oxygen in water at 25C and at 75C and 1 atm.
-
An S-590 alloy component (Figure) must have a creep rupture lifetime of at least 100 days at 500(C (773 K). Compute the maximum allowable stress level. 10 T(20 + log 1,XR-h) 25 30 35 40 45 50 1000...
-
Allocated Company is Woolworths, Need to use Woolworths financial report 2022. https://www.woolworthsgroup.com.au/content/dam/wwg/investors/reports/2022/full-year/2022%20Annual%20Report.pdf Calculate...
-
A financial manager is an employee_ who helps not-for-profit organizations account for all donations and expenditures who invests a company's profits in order to generate more income for the firm...
-
The A-Rod Company hs not been receiving as much from its customer base as in prior years. There is concern that this will hamper future cash flows. The firm had annual sales of $18,000,000. It does...
-
(4) Tim has also received another offer from a Canadian Basketball club in the Alberta province. The club provides him with a three-year contract with an annual salary of 520,000 Canadian dollars...
-
QUESTION 9 T1 - step 3 - line 34 This is the allowable RRSP deduction Anna can claim, according to the Schedule 7 form QUESTION 10 S8 - part 3 - line 7 OR 9 The amount of CPP contributions Anna is...
-
Refer to the information in PB2-3 for Knight Company. Required: 1. Prepare a journal entry showing the transfer of Job 102 into Finished Goods Inventory upon its completion. 2. Prepare the journal...
-
Why is disclosure of depreciation or amortization methods and rates so important?
-
Use MuPAD to compute the following limits. x + 1 lim x- 00 . 3x b. lim 2x 2x + 3 X -00
-
Find the expression for the sum of the geometric series for r 1. n-1 k=0
-
A particular rubber ball rebounds to one-half its original height when dropped on a floor . a. If the ball is initially dropped from a height h and is allowed to continue to bounce, find the...
-
Minden Company is a wholesale distributor of premium European chocolates. The company s balance sheet as of April 3 0 is given below: Minden Company Balance Sheet April 3 0 Assets Cash $ 1 1 , 4 0 0...
-
What is the effect on substantive tests of accounts payable and related expenses when the auditor assesses the risk of material misstatement as low because the client has effective internal controls?
-
You will reflect on your personal history with conflict. I am looking for: (a) your critical understanding of course material (COM 3350: Conflict Resolution); and (b) your ability to apply that...

Study smarter with the SolutionInn App


