Question:
You plan to build a Koi pond in your backyard but worry about how long the fish can survive if the pond freezes over. Without access to new supplies of oxygen, what would happen? Suppose midwinter temperatures hit a low of \(-10^{\circ} \mathrm{C}\) over an extended period of time. If your pond is 1 meter deep, chances are it remains at a constant temperature \(10^{\circ} \mathrm{C}\) at the bottom. If the freezing process excludes oxygen from the ice and forces it into the remaining water, the fish could survive longer. How much ice would need to be formed to increase the oxygen content of the water by \(10 \%\) ? How long would the weather have to remain cold to do that? You can use the material properties for water given in the example in Section 6.5.3.
Transcribed Image Text:
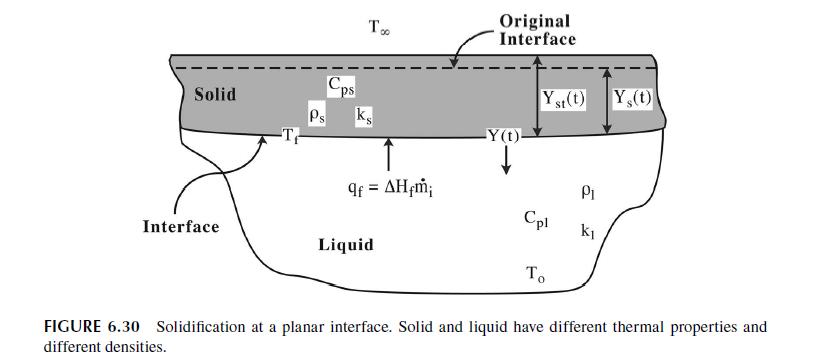
6.5.3 MELTING AND SOLIDIFICATION [6,10] Our discussion of diffusion with instantaneous reaction on the boundary provides the perfect intro- duction for a discussion of the next, important transport process involving a moving boundary: melting or solidification. In this specific example, we will be discussing the solidification of a liquid, but the corresponding melting problem can be treated in the same way. Consider the surface of a lake or pond shown in Figure 6.30. We suppose that the liquid originally fills the entire space, y > 0, and is being solidified from the exposed surface by removal of heat. The exposed surface must be at a temperature below the freezing point, Ty, and we will assume that the upper surface is held at a constant temperature equal to the ambient air temperature, T. The liquid is initially at a uniform temperature above its freezing point, T, and remains at that temperature far away from the surface. We begin our analysis by considering control volumes located within the solid and liquid. These are shown in Figure 6.31. In both the solid and liquid, we assume heat transfer by conduction only, with no generation. This is a somewhat restrictive assumption. Convection in the liquid will limit how fast solidification can occur. That is why moving water freezes more slowly. If we write an energy balance about the control volumes, assuming one-dimensional conduction, we arrive at two partial differential equations identical in form to equation (6.135). aT T = t = aT, T at = ki PjCpl (6.210) ks s = (6.211) PsCps









