Analyze the frame in Figure P11.30 by moment distribution. Determine all reactions and draw the shear and
Question:
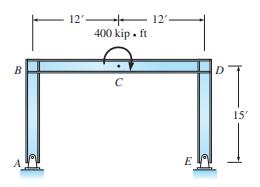
Analyze the frame in Figure P11.30 by moment distribution. Determine all reactions and draw the shear and moment curves. Sketch the deflected shape. \(E\) is constant and equals 30,000 kips/in. \({ }^{2}\).
Transcribed Image Text:
B 12 H + 400 kip. ft C 12 E 15'
Step by Step Answer:

This question has not been answered yet.
You can Ask your question!
Related Book For 

Fundamentals Of Structural Analysis
ISBN: 9780073398006
5th Edition
Authors: Kenneth Leet, Chia-Ming Uang, Joel Lanning
Question Posted:
Students also viewed these Engineering questions
-
A system has the following differential equation: Determine Φ(t) and its transform Φ(s) for the system. 0 0 2
-
Solve each polynomial inequality in Exercises 142 and graph the solution set on a real number line. Express each solution set in interval notation. x = 4x 2
-
Write a Java program to assist the sales manager to review quotation detail. The GUI program works as follow. Upon start up, only the Load button is enabled to allow user to load the quotation items...
-
Advertising plays a major role in the ______________ stage of the product life cycle, and_______________ plays a major role in the maturity stage.
-
Replace the loading on the frame by a single resultant force. Specify where its line of action intersects member AB, measured from A. Given: F1 = 500 N a = 3 m F2 = 300 N b = 2 m c = 1 m F3 = 250 N d...
-
Two 3.0 g point charges on 1.0-m-long threads repel each other after being equally charged, as shown in FIGURE CP22.73. What is the magnitude of the charge q? 1.0 m /20 20 \1.0 m 3.0 g 3.0 g FIGURE...
-
How to establish the terms of the attorney-client agreement?
-
On November 1, 2015, Aviation Training Corp. borrows $60,000 cash from Community Savings and Loan. Aviation Training signs a three-month, 7% note payable. Interest is payable at maturity. Aviations...
-
Refer to point 1 entitled "repair of damaged harbour wall". Discuss how the R132 million would be recognised in the financial statements of SSSS as at 30 September 2012. You should apply the relevant...
-
Analyze the Vierendeel truss in Figure P11.31 by moment distribution. Draw the shear and moment curves for members \(A B\) and \(A F\). Sketch the deflected shape, and determine the deflection at...
-
Analyze the frame in Figure P11.29 by moment distribution. Draw the shear and moment curves. Sketch the deflected shape. \(E\) is constant and equals \(30,000 \mathrm{kips} / \mathrm{in} .{ }^{2}\)....
-
Consider the market for butter, shown in the accompanying figure. The government imposes an excise tax of $0.30 per pound of butter. What is the price paid by consumers post-tax? What is the price...
-
Wildhorse Company has a unit selling price of $720, unit variable costs of $380, and fixed costs of $196,520. Compute the break-even point in sales units using (a) the mathematical equation and (b)...
-
On 1 January 2019 the carrying value of equipment was R19 720 (cost price, R25 670 and accumulated depreciation, R5 950). On 1 January 2019 equipment costing R5 670 was purchased (not included in the...
-
AT-bill with a face value $10,000 and 120 days to maturity is selling at a bank discount ask yield of 2%. a. What is the price of the bill? b. What is its bond equivalent yield?
-
a. If =2.5, find P(X = 8). c. If = 0.5, find P(X = 3).
-
1.Is the use of AI technology for work automation unethical? Why or why not? 2.Is the use of AI technology for work automation unethical? Why or why not? 3.Why should businesses and governments find...
-
Fully vested incentive stock options exercisable at $50 per share to obtain 24,000 shares of common stock were outstanding during a period when the average market price of the common stock was $60...
-
Write the expression in radical notation. Then evaluate the expression when the result is an integer. 23 -1/2
-
The effective molecular mass of air is defined as the mass of a kmol of elementary particles of which 78.09% are nitrogen molecules, 20.95% are oxygen molecules, 0.933% are argon atoms, and 0.027%...
-
A gallon of gasoline has a mass of about 3.0 kg. Further, a kg of gasoline has an energy content of about 45,500 kJ/kg. If an experimental automobile requires just 10. kW of power to overcome air...
-
Determine the value of the stoichiometric coefficients for the combustion of an oil (assumed molecular formula CH 2 ) in oxygen: CH 2 + aO 2 = bCO 2 + cH 2 O. Confirm your answer is correct! (A: a =...
-
The information for Bland Media Company at the end of the fiscal year, June 30, 20X8 are listed in this spreadsheet . Additional information: Capital were $145000 at May 1, 20X7, additional capital...
-
Budget Problem Solutions 1. Sales budget: $ 9,500,000 2. schedule of expected cash collections: $9,482,500 2. A purchases budget: $5,377,500 3. Cash payment schedule: $5,362,500 4. A cash budget: $...
-
Required: Prepare the following budgets and schedules for the year, showing both quarterly and total amounts. 1. Sales budget and a schedule of expected cash collections. 2. A purchases budget 3....

Study smarter with the SolutionInn App


