Determine the reactions of structure 10 kips 5 kips/ft 4' B
Question:
Determine the reactions of structure
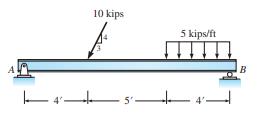
Transcribed Image Text:
10 kips 5 kips/ft 4' B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
EM 0 0 A60 A 6 1F 0 Ax A A F ...View the full answer

Answered By

Lisper Wanja
I am an experienced and highly motivated writer with a passion for the skills listed. I have a proven track record of my expertise and my aim is to deliver quality, well-detailed and plagiarism free projects. My genuine passion for writing combined with my ongoing professional development through school and research makes me an ideal candidate within for any assignment.
4.90+
233+ Reviews
388+ Question Solved
Related Book For 

Fundamentals Of Structural Analysis
ISBN: 9780073398006
5th Edition
Authors: Kenneth Leet, Chia-Ming Uang, Joel Lanning
Question Posted:
Students also viewed these Engineering questions
-
The rates of many atmospheric reactions are accelerated by the absorption of light by one of the reactants. For example, consider the reaction between methane and chlorine to produce methyl chloride...
-
With current spectroscopic techniques (discussed in Chapters 1517), chemists are generally able to determine the structure of an unknown organic compound in just one day. These techniques have only...
-
Find the point on the plane z = x + y + 1 closest to the point P = (1, 0, 0). Minimize the square of the distance.
-
Consider the case of Rutherford scattering in the event that m1 >> m2. Obtain an approximate expression for the differential cross section in the LAB coordinate system.
-
CarryALL, Inc., makes and sells small cargo trailers to individuals and small businesses. Since its opening in 1990, it has allocated indirect costs (IDC) to its three manufacturing plants based on...
-
What do you know about the mathematical value of the annual worth of a project under each of the following conditions? a. The present worth of the project is greater than 0 . b. The present worth of...
-
During 2011, Rooster Company purchased 5,000 shares of Hen Company common stock for $18 per share and 3,200 shares of Egg Company common stock for $21 per share. These investments are intended to be...
-
There are two primary reasons that guide my approach to back up files externally. First, there's the aspect of cost savings. Investing in a hard drive with sufficient storage capacity constitutes a...
-
Determine the reactions of structure 15 kip. ft 5 5' B D 12] E 8 kips 6 kips 10'
-
Consider the building in Figure P2.22, which has a width into the page of \(35 \mathrm{ft}\). Maximum inundation height, \(h_{\text {max }}\), and flow velocity, \(u_{\max }\), have been determined...
-
Consider the following large organizations. Would you classify them as primarily cost leaders, product differentiators, or focused competitors according to the Porter Strategy Framework?Why? Dominant...
-
Use the following information to determine the adjustments, if any, to ICO. (Hint: if you are adjusting ICO, should the adjustments be pre-tax or net of tax?) If you need to increase ICO, enter your...
-
2. After taking her first course in business mathematics, Alyssa is interested in purchasing a 180-day T-Bill with a face value of $20,000 and a rate of 3.5% p.a. (6 points) a) Calcule le prix...
-
Clara is a 14-year-old student presenting significant academic delays as compared to her peer group. She has just arrived in our community and has just been enrolled in school with no accompanying...
-
In one hundred words or less, define the theories of Lloyd Bitzer (rhetorical exigence) and Sidney Jourard (self-disclosure) and explain a situation where a rhetorical exigence required that YOU...
-
Your Company enters into a finance lease as the lessee and determines the present value of the lease payments and residual value is $500,000. Your Company also incurs $8,000 in legal fees to execute...
-
Refer to the data in Exercise 14-24. The division manager learns that he has the option to lease the asset on a year-to-year lease for $148,000 per year. All depreciation and other tax benefits would...
-
Represent each of the following combination of units in the correct SI form using an appropriate prefix: (a) m/ms, (b) k m, (c) k s /mg, and (d) k m N.
-
Write the charge balance for a solution of H 2 SO 4 in water if H 2 SO 4 ionizes to HSO 4 and SO 2 4 - .
-
For a 0.1 M aqueous solution of sodium acetate, Na + CH 3 CO - 2 , one mass balance is simply [Na + ] = 0.1 M. Write a mass balance involving acetate.
-
(a) Following the example of Mg(OH) 2 in Section 7-5, write the equations needed to find the solubility of Ca(OH) 2 . Include activity coefficients where appropriate. Equilibrium constants are in...
-
What is the essence of conflict negotiation, and how do different conflict negotiation models, such as integrative, distributive, and principled negotiation, provide distinct approaches for resolving...
-
Who are the staff members from whom you would collect useful information and requirements of store keeper's job?
-
Y5 Question 2. Formulate and explain the duality result between indirect utility and expenditure of a consumer with rational, continuous and strictly convex preferences. [12.5 marks]

Study smarter with the SolutionInn App


