Determine the reactions of structure 1.2 kips/ft B A 18k 12'- D 9
Question:
Determine the reactions of structure
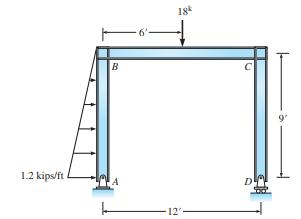
Transcribed Image Text:
1.2 kips/ft B A 18k 12'- D 9
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
912 9186D 120 D ...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Structural Analysis
ISBN: 9780073398006
5th Edition
Authors: Kenneth Leet, Chia-Ming Uang, Joel Lanning
Question Posted:
Students also viewed these Engineering questions
-
Problem 2. Based on a previous midterm exam problem. This problem asks you to analyze the effect of using a "bumpless" feedback control implementation (Figure 1). C2 + A- C3 Figure 1: "Bumpless"...
-
The rates of many atmospheric reactions are accelerated by the absorption of light by one of the reactants. For example, consider the reaction between methane and chlorine to produce methyl chloride...
-
With current spectroscopic techniques (discussed in Chapters 1517), chemists are generally able to determine the structure of an unknown organic compound in just one day. These techniques have only...
-
In this exercise, we prove that for all x, y 0: a + b 1 1.40 y B where a 1 and 1 are numbers such that a +B- = 1. To do this, we prove that the function f(x,y) = ax + _xy xy satisfies f(x, y) 0 for...
-
It is found experimentally that in the elastic scattering of neutrons by protons (mn mp) at relatively low energies, the energy distribution of the recoiling protons in the LAB system is constant up...
-
Calculate O2 mixture (298 15 K, 1 bar) , for oxygen in air, assuming that the mole fraction of O 2 in air is 0.210. Use the conventional molar Gibbs energy defined in Section 6.17.
-
Recall, even if evidence is relevant under the general relevance test, there may be special rules that preclude the introduction of the evidence. In addition to a persons character or habits, what...
-
The franchise arrangement between McDonalds and its franchisees is summarized in the following note from McDonalds 2007 annual report. Individual franchise arrangements generally include a lease and...
-
Adjusting Entries are necessary because of one accounting basis and two accounting principles. Which basisof accounting, Cash or Accrual, makes adjusting entries necessary and why does it make...
-
Determine the reactions of structure BO 1 kip. ft 12' 10'
-
Determine the reactions of structure B -3m- .3m 4m D 20 kN 15 KN
-
The Alexander Company reported the following income statement for 2013: Assume that all depreciation and 75 percent of the firm's selling, general, and administrative expenses are fixed costs and...
-
Cherokee Incorporated is a merchandiser that provided the following information: Number of units sold Selling price per unit Variable selling expense per unit Variable administrative expense per unit...
-
Many colleges and universities are supported by the government, while others are supported by private organizations. What bearing does the source of support have on the determination of authoritative...
-
How could variance analysis be employed to assess the discrepancies between the projected surplus in the 2 0 2 4 2 0 2 5 fiscal year and the subsequent shortfalls in t 2 . What proactive measures...
-
solve the following Question Completion Status: Required: 1. Using Rapala's financial information prepare the Contribution Income Statement. 2. Calculate the contribution margin. 3. Calculate the...
-
What would be the journal entries for these statements December business transactions Dec 1 - Collected $ 7 1 4 from Franklin from a previous engagement in Oct. Dec, 2 - Paid $ 1 , 5 1 8 on Gusto's...
-
Julie Whiteweiler made $930 this week. Only social security (fully taxable) and federal income taxes attach to her pay. Whiteweiler contributes $100 each week to her company's 401(k) plan and has $25...
-
l ask this second time correnct answer is 38,01 can we look pls Consider a non-conducting rod of length 8.8 m having a uniform charge density 4.5 nC/m. Find the electric potential at P, a...
-
(a) Write the mass balance for CaCl 2 in water if the species are Ca 2+ and Cl - . (b) Write the mass balance if the species are Ca 2+ , Cl - , CaCl - , and CaOH + . (c) Write the charge balance for...
-
Write the charge and mass balances for dissolving CaF 2 in water if the reactions are CaF,(8) = Ca2+ + 2F Ca?+ + H,O = CAOH+ + H* Ca2+ + F = CaF+ CaF2(s) = CaF2(aq) F + H* = HF(aq) HF(aq) + F = HF,
-
Write charge and mass balances for aqueous Ca 3 (PO 4 ) 2 if the species are Ca 2+ , CaOH + , CaPO - 4 , PO 3- 4 , HPO 2 4 - , H 2 PO - 4 , and H 3 PO 4 .
-
Decision-making involves choosing from among alternatives. A number of factors help influence whether decision making will be centralized or decentralized, including company size, amount of capital...
-
In a lawsuit happy society, everyone is to blame. But are they? Consider the case where a patient does not follow doctor's orders for wound care (i.e., difficult to prove) and acquires a...
-
Physician-assisted euthanasia (PAE) has become legal in some states (OR, WA, MT, VT, etc.). Discuss reasons why all 50 states have not legalized PAE. List the legal and ethical concepts in support of...

Study smarter with the SolutionInn App


