Consider the combination of the two heat engines, as in Fig. P5.4. How should the intermediate temperature
Question:
Consider the combination of the two heat engines, as in Fig. P5.4. How should the intermediate temperature be selected so that the two heat engines have the same efficiency, assuming Carnot cycle heat engines.
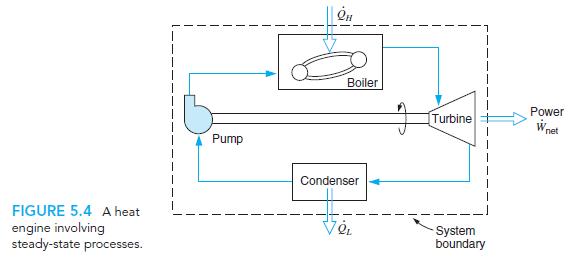
Transcribed Image Text:
Boiler Power Turbine W net Pump Condenser FIGURE 5.4 A heat engine involving steady-state processes. System boundary
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (4 reviews)
The intermediate temperature should be selected so that ...View the full answer

Answered By

Firoz K
I have extensive experience in education and tutoring, having worked as a tutor for the past three years in both group and individual settings. During my time as a tutor, I have successfully helped students improve their academic performance in a variety of subjects, including mathematics, science, language arts, and social studies. I have also developed and implemented personalized learning plans and differentiated instruction techniques to accommodate the individual needs of my students. Moreover, I have effectively communicated with parents and teachers to ensure that the students receive the best possible education and guidance. My strong organizational, communication, and problem-solving skills have enabled me to successfully collaborate with students, parents, and teachers in order to provide an effective and enjoyable learning experience.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
Consider the combination of capacitors in figure.(a) What is the equivalent capacitance of the group?(b) Determine the charge on eachcapacitor. 24.0 F 2.00 F 4.00 F 36.0 V 8.00 uf
-
Consider the combination of a heat engine and a heat pump, as given in Problem 5.41, with a low temperature of 720 R. What should the high temperature be so that the heat engine is reversible? For...
-
Consider two Carnot heat engines operating in series. The first engine receives heat from the reservoir at 1800 K and rejects the waste heat to another reservoir at temperature T. The second engine...
-
In figure, one end of a uniform beam weighing 20 x V3N is attached to a wall with a hinge. The other end is supported by a wire connected to the wall as shown. If the tension in the wire is a x 10N,...
-
What is the value of the mean score for any standard normal distribution? What is the value of the standard deviation for any standard normal distribution? Explain why this is true for any standard...
-
In the evaluation of the symmetry content of a morphological crystal, you can make a list of all of the symmetry elements or you can use an internationally accepted shorthand notation. Which of these...
-
To keep blood from pooling in their lower legs on plane trips, some people wear compression socks. These socks are sold by the pressure they apply; a typical rating is 20 mm Hg. Over what vertical...
-
1. What is the responsibility of management and the auditor with respect to the internal controls of a client? 2. Groupon disclosed a material weakness in its internal controls saying that it had...
-
Use the Black-Scholes model to price a European call option. Suppose you calculated N(d;) = 0.4771 and N(d2) = 0.3968. If the current stock price is $50, the strike price is $52, the risk-free rate...
-
KeyCite this case: United States v. Martinez-Jimenez, 864 F.2d 664 (9th Cir. 1989), what are the citations of the 1995 and 1993 cases listed under Positive History that have a yellow flag? For...
-
The air conditioner in the previous problem is turned off. How quickly does the house heat up in degrees per second (C/s)? Data from previous problem An air conditioner on a hot summer day removes 8...
-
Consider a combination of a gas turbine power plant and a steam power plant, as shown in Fig. P5.4. The gas turbine operates at higher temperatures (thus called a topping cycle) than the steam power...
-
Gonzales Upholstery, Inc.'s trial balance at the end of its fiscal year follows. The following information is also available: a. A study of the company's insurance policies shows that $380 is...
-
What impact does the Security and Exchange Commission's regulations have on corporate governance? How it can better the governance?
-
Businesses can assess their financials using a balance sheet. For property and casualty insurance companies, there are three major sections of the balance sheet: assets, liabilities, and...
-
Charlie had an account balance of $8,500 on April 1. He received a payment of $30,000 on April 15 from a sale of an investment. On April 29, he purchased a 32000 GIC for a 3 year term. The bank pays...
-
Velma, 37, is a target group member for the Work Opportunity Tax Credit (WOTC). In 2022, she was hired and worked 200 hours, earning $3,500. What is the amount of the WOTC credit her employer can...
-
Evaluate the different approaches accounting to forecasting income income for the investment?
-
Repeat Problem 42 if the eyepiece location is adjusted slightly so that the final image is at the viewer's near point (25.0 cm) instead of at infinity.
-
Consider the activities undertaken by a medical clinic in your area. Required 1. Do you consider a job order cost accounting system appropriate for the clinic? 2. Identify as many factors as possible...
-
In each case, identify the most likely position at which monobromination would occur. (a) (b) (c) (d) N.
-
Identify the carboxylic acid and the alcohol that are necessary in order to make each of the following compounds via a Fischer esterification: a. b. c. CH 3 CH 2 CO 2 C (CH 3 ) 3
-
Determine the structures of compounds A through F: Na,Cr,0, H2SO4, H20 [H'] A EtO soch 1) LIAI(OR)H `2) H20 xS NH3
-
SageGreen can claim the Advanced manufacturing production credit (section 45X) by strategically investing in the production and selling of eligible solar components. The new Section 45X provides a...
-
Access DRL's annual report and 20-F for the 2019-20 fiscal year on the Investor Relations section of the company's website (www.drreddys.com/investors/reports-and-filings). Answer the following...
-
Describe Kroger's accumulated earnings for each of the 2 years and the meaning of the accumulated earnings account on the balance sheet. Determine the par value of the common shares based on Kroger's...

Study smarter with the SolutionInn App


