In a jet engine a flow of air at 1000 K, 200 kPa, and 30 m/s enters
Question:
In a jet engine a flow of air at 1000 K, 200 kPa, and 30 m/s enters a nozzle, as shown in Fig. P4.23, where the air exits at 850 K, 90 kPa. What is the exit velocity, assuming no heat loss?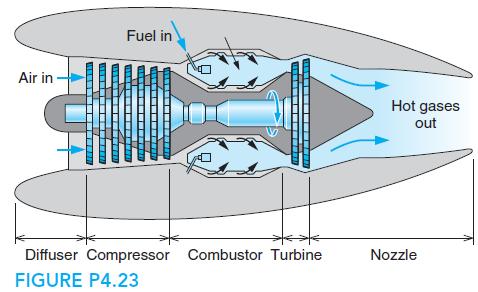
Transcribed Image Text:
Fuel in Air in Hot gases out Diffuser Compressor Combustor Turbine Nozzle FIGURE P4.23
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 70% (10 reviews)
To determine the exit velocity of the air in the nozzle we can use the principle of conservation of ...View the full answer

Answered By

Akshay Shete
I have extensive experience as a tutor, both online and in-person. I have worked with students of all ages and abilities, and am skilled at adapting my teaching style to meet the needs of each individual student. I have a strong background in a variety of subjects, including math, science, and English, and am able to break down complex concepts in a way that is easy for students to understand. In addition to my subject matter expertise, I am also a patient and supportive teacher, and am committed to helping my students succeed. Whether I am working with a struggling student who needs extra help to catch up, or an advanced student looking to get ahead, I am able to provide the guidance and support they need to reach their goals. Overall, my hands-on experience as a tutor has prepared me to be a confident and effective teacher, and I am excited to use my skills to help students succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
In a jet engine a flow of air at 1800 R, 30 psia and 90 ft/s enters a nozzle where the air exits at 1500 R, 13 psia, as shown in Fig. P.6.33. What is the exit velocity assuming no heat loss?
-
The exit nozzle in a jet engine receives air at 1200 K, 150 kPa with neglible kinetic energy. The exit pressure is 80 kPa and the process is reversible and adiabatic. Use constant heat capacity at...
-
The turbine section in a jet engine receives gas (assume air) at 1200 K, 800 kPa with an ambient atmosphere at 80 kPa. The turbine is followed by a nozzle open to the atmosphere and all the turbine...
-
In Problems 2738, the reduced row echelon form of a system of linear equations is given. Write the system of equations corresponding to the given matrix. Use x, y; or x, y, z; or x 1 , x 2 , x 3 , x...
-
What operations must be used to solve an applied problem if all of the parts but one are given and the total of all the parts is given? Write an example.
-
Indicate the limiting reactant shown in the following illustration: ZnS(s) S (s)suz (s)S + (s)uz
-
What is the structure of hemoglobin? Which part of the hemoglobin molecule binds to oxygen?
-
The stockholders equity of Pondside Occupational Therapy, Inc., on December 31, 2011, follows: On April 30, 2012, the market price of Pondsides common stock was $11 per share and the company...
-
Consider different Internet security approaches and answer the following: a) Explain an advantage of application-level security (versus transport-level security). b) Explain an advantage of...
-
The before-tax income for Hawks Corp. for 2019 was $ 115,500 ; for 2020, it was $ 79,000 . However, the accountant noted that the following errors had been made: 1. Sales for 2019 included $ 39,300...
-
A diffuser receives 0.1 kg/s steam at 500 kPa, 350C. The exit is at 1 MPa, 400C with negligible kinetic energy and the flow is adiabatic. Find the diffuser inlet velocity and the inlet area.
-
Superheated vapor ammonia enters an insulated nozzle at 30C, 1000 kPa, as shown in Fig. P4.25,with a low velocity and at a rate of 0.01 kg/s. The ammonia exits at 300 kPa with a velocity of 450 m/s....
-
Consider the glider of Example 7.7 (Section 7.2) and Fig. 7.16. As in the example, the glider is released from rest with the spring stretched 0.100 m. What is the displacement x of the glider from...
-
Discuss FOUR (4) principles that have a strong, sustainable and positive reputation of brand. Provide ONE (1) relevant example for each principle. You must include the references.
-
How does Data analytics impact the field of communication? Describe a current or emerging trend. Identify the business need or communication problem the trend resolves. Explain the current or...
-
Provide two specific examples of how people are using or abusing mass media laws today. Consider the ethical implications as well the legal. Explain the laws that are being violated. Would you...
-
You're the human resources manager at the CSI Business Hotel in Chicago. You have an opening for a Guest Services Manager. Hoping for more customers by the summer, you know you will need to go all...
-
What are strategies adjustment messages that salvage customers' trust and promote further business?
-
For each of the following pairs of SN2 reactions, indicate which reaction occurs faster: a. CH3CH2Br + H2O or CH3CH2Br + HO b. c. d. CH3CH2Cl + I or CH3CH2Br + I CH3CHCH2BHO or CH CH2CHBrHO CH CH,CI...
-
Identify the tax issues or problems suggested by the following situations. State each issue as a question. Jennifer did not file a tax return for 2007 because she honestly believed that no tax was...
-
A compressor receives R-134a at 10oC, 200 kPa with an exit of 1200 kPa, 50oC. What can you say about the process?
-
A supply of 5 kg/s ammonia at 500 kPa, 20C is needed. Two sources are available one is saturated liquid at 20C and the other is at 500 kPa and 140C. Flows from the two sources are fed through valves...
-
A counter flowing heat exchanger has one line with 2 kg/s at 125 kPa, 1000 K entering and the air is leaving at 100 kPa, 400 K. The other line has 0.5 kg/s water coming in at 200 kPa, 20C and leaving...
-
Consider three Treasury bonds with 10 years to maturity. The three bonds differ only in their coupon rates0%, 2%, and 4%all of which are compounded semi-annually. Plot the relationship between price...
-
The fixed-charge coverage ratio requirements limits a company's ability to Multiple choice question. repurchase stock. enter into a merger with another company. pay off other debts. pay dividends.
-
Starbucks' decision to buy out its joint venture partner in Japan following several years of operation and instead establish a wholly owned subsidiary Multiple Choice suggests that the company no...

Study smarter with the SolutionInn App


