In Fig. P3.5, CV A is the mass inside a piston/ cylinder, and CV B is the
Question:
In Fig. P3.5, CV A is the mass inside a piston/ cylinder, and CV B is the mass plus the piston outside, which is the standard atmosphere. Write the energy equation and work term for the two CVs, assuming we have a nonzero Q between state 1 and state 2.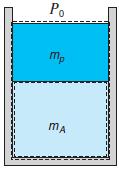
Transcribed Image Text:
Po mp
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (10 reviews)
The energy equation for the two control volumes can be written as CV A U Q WA 0 CV B U Q WB 0 where ...View the full answer

Answered By

Akshay Shete
I have extensive experience as a tutor, both online and in-person. I have worked with students of all ages and abilities, and am skilled at adapting my teaching style to meet the needs of each individual student. I have a strong background in a variety of subjects, including math, science, and English, and am able to break down complex concepts in a way that is easy for students to understand. In addition to my subject matter expertise, I am also a patient and supportive teacher, and am committed to helping my students succeed. Whether I am working with a struggling student who needs extra help to catch up, or an advanced student looking to get ahead, I am able to provide the guidance and support they need to reach their goals. Overall, my hands-on experience as a tutor has prepared me to be a confident and effective teacher, and I am excited to use my skills to help students succeed.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Fundamentals Of Thermodynamics
ISBN: 9781118131992
8th Edition
Authors: Claus Borgnakke, Richard E. Sonntag
Question Posted:
Students also viewed these Sciences questions
-
A piston can freely move inside a horizontal cylinder closed from both ends. Initially, the piston separates the inside space of the cylinder into two equal parts each of volume V0, in which an ideal...
-
A frictionless piston - cylinder device contains 5 kg of nitrogen at 100 kPa and 250 K. Nitrogen is now compressed slowly according to the relation PV1.4 5 constant until it reaches a final...
-
A piston cylinder contains 1 kg water at 20oC with a constant load on the piston such that the pressure is 250 kPa. A nozzle in a line to the cylinder is opened to enable a flow to the outside...
-
The inequality describes the range of monthly average temperatures T in degrees Fahrenheit at a certain location. (a) Solve the inequality. (b) If the high and low monthly average temperatures...
-
Construct a bar graph showing total sales by salesperson for Happy's Gift Shoppe in Table 7-7.
-
Would it be more or less likely that cells of the kind we know would evolve on a gas giant such as the planet Jupiter?
-
A study divided college students into two groups. One group listened to Mozart or other classical music before being assigned a specific task and the other group simply was assigned the task without...
-
On January 1, 2008, Johnson Corporation granted 5,000 options to executives. Each option entitles the holder to purchase one share of Johnsons $5 par value common stock at $50 per share at any time...
-
7. What is the output of the following program? int a = 10; void main() 8. { int a 20; } cout < < a < < ::a; Consider the following program: int main() int k = 0; int j = 0; while (k == j) } int k;...
-
Soft selling occurs when a buyer is skeptical of the quality or usefulness of a product or service. For example, suppose youre trying to sell a company a new accounting system that will reduce costs...
-
If a process in a control mass increases energy E 2 E 1 > 0, can you say anything about the sign for 1Q 2 and 1W 2 ?
-
A500-W electric space heater with a small fan inside heats air by blowing it over a hot electrical wire. For each control volume (a) Wire only, (b) All the room air, (c) Total room air plus the...
-
Selected information from Loblaw Companies Limiteds income statements for three recent years follows (dollars in millions): Instructions a. Calculate gross profit, and profit from operations, for...
-
In what respects is native freehold land in Cook Islands more limited than common law freehold in fee simple?
-
Find the Big-Oh estimation of the following a. f(n)=(n-2)4+ n(n+3), f(n) = O(................) b. g(n)= n(n+1)(2n+3)/2, g(n) =O.............. c. k(n)= nlog( n +2n) + n(n+1), k(n)=O(.....................
-
What approaches can leaders take to navigate through ambiguity and uncertainty, fostering agility and resilience within their teams to thrive in dynamic and unpredictable business environments?
-
Consider the following model with two assets and three states, and suppose that r = 0%. n 1 2 Sn (0) 3 9 Sn(1,w) Sn(1, W2) Sn(1, W3) 4 2 11 5 3 8 Show that there exists a portfolio H which is a...
-
Your client, Dean, is very excited to be buying his first home. As he prepares for his escrow closing, he asks you what the last step is before the process is complete?
-
How could IR spectroscopy distinguish among 1-hexyne, 2-hexyne, and 3-hexyne?
-
Ashlee, Hiroki, Kate, and Albee LLC each own a 25 percent interest in Tally Industries LLC, which generates annual gross receipts of over $10 million. Ashlee, Hiroki, and Kate manage the business,...
-
A piston/cylinder has 0.5 kg air at 2000 kPa, 1000 K as shown. The cylinder has stops so Vmin = 0.03 m3. The air now cools to 400 K by heat transfer to the ambient. Find the final volume and pressure...
-
The air conditioner in a house or a car has a cooler that brings atmospheric air from 30oC to 10oC both states at 101 kPa. If the flow rate is 0.5 kg/s find the rate of heat transfer.
-
A flow of liquid glycerine flows around an engine, cooling it as it absorbs energy. The glycerine enters the engine at 60oC and receives 9 kW of heat transfer. What is the required mass flow rate if...
-
20 3 A = and B = 5 7 6-1 (a) Find AB if possible. (b) Find BA if possible. (c) Find A if possible. (Hint: A = A.A.)
-
1. Let A= -1 2 B= 2 1 52 13 D= 1 31 01 E= 3 24 -112 5 13 Evaluate each of the following expressions or explain why it is undefined. (a) (3 points) DT (AET)-4(ED)T (b) (2 points) det(C) (c) (2 points)...
-
Write a 1050-1400 word paper in which you examine clinical psychology Address the following items: - Discuss the history and evolving nature of clinical psychology - Explain the role of research and...

Study smarter with the SolutionInn App


