(A) Are the following pairs of molecules isomers? (B) Are the pairs of molecules represented by the...
Question:
(A) Are the following pairs of molecules isomers?
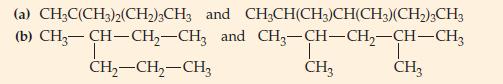
(B) Are the pairs of molecules represented by the following structural formulas isomers?
Transcribed Image Text:
(a) CH3C(CH3)2(CH₂)3CH3 (b) CH3-CH-CH₂-CH3 CH₂-CH₂-CH3 and CH₂CH(CH3)CH(CH3)(CH₂)3CH3 and CH3-CH-CH₂-CH-CH3 T CH3 I CH3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
A a No the two molecules are not isomers They have the same molecular ...View the full answer

Answered By

Milan Mondal
I am milan mondal have done my Msc in physics (special astrophysics and relativity) from the University of burdwan and Bed in physical science from the same University.
From 2018 I am working as pgt physics teacher in kendriya vidyalaya no2 kharagpur ,west bengal. And also I am doing advanced physics expert in chegg.com .also I teach Bsc physics .
I love to teach physics and acience.
If you give me a chance I will give my best to you.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Are the following pairs of molecules isomers or not? (a) CHCH(CH3)(CH)3CH3 (b) CH-CH-CH-CH3 CH-CH3 and CH3CHCH(CH3)(CH)3CH3 and CH-CH-CH-CH-CH CH3
-
Examine the relationships of isomers as described in Figure 2.8 (p. 56). Then classify the following pairs of structures as structural isomers, conformers, configurational (cis-trans) isomers, or...
-
How could you distinguish between the following pairs of isomers by 1H NMR spectroscopy? a. CH3CCl3 and CH2ClCHCl2 b. CH3CH2CH2OH and (CH3)2CHOH c d. 0 C-OCH and H C OCH2CH CH2-CH=O and C CH
-
In Exercises verify that the function satisfies the differential equation. Function y = 2 sin x + 3 Differential Equation y"+y = 3
-
Porter has a capital balance of $420,000 after adjusting assets to fair market value. Billings contributes $200,000 to receive a 30% interest in a new partnership with Porter. Determine the amount...
-
Explain each of the following statements: a. The Wagner Act of 1935 reduced the costs of providing union services and thereby increased the number of union members. b. The Wagner Act of 1935...
-
A vertical jet of water leaves a nozzle at a speed of \(10 \mathrm{~m} / \mathrm{s}\) and a diameter of \(20 \mathrm{~mm}\). It suspends a plate having a mass of \(1.5 \mathrm{~kg}\) as indicated in...
-
The following condensed income statements of the Jackson Holding Company are presented for the two years ended December 31, 2018 and 2017: On October 15, 2018, Jackson entered into a tentative...
-
Crane reported the following results from the sale of 5000 units in May: sales $300000, variable costs $165000, fixed costs $65000, and net income $70000. Assume that Crane increases its selling...
-
Determine the mass, in grams, of (a) 2.10 x 10 2 mol S 8 ; (b) 5.02 x 10 22 molecules of palmitic acid, C 16 H 32 O 2 ; (c) A quantity of the amino acid histidine, C 6 H 9 N 3 O 2 , containing 2.95...
-
Determine the number of moles of (a) N 2 O 4 in a 115 g sample; (b) N atoms in 43.5 g of Mg(NO 3 ) 2 ; (c) N atoms in a sample of C 7 H 5 (NO 2 ) 3 that has the same number of O atoms as 12.4 g C 6 H...
-
Wally Los Gatos, owner of Wally's Wonderful World of Wallcoverings, Etc., has hired you as a consultant to design a database management system for his new online marketplace for wallpaper, draperies,...
-
You observe a stock price of $18.75. You expect a dividend growth rate of 5% and the most recent dividend paid was $1.50. What is the "implied" required return?
-
Two lives aged x and y take out a policy that will pay out 15,000 on the death of (x) provided that (y) has died at least 5 years earlier and no more than 15 years earlier. (1) Express the present...
-
11. Solve using quadratic formula a) 4x + 6(4-x) = 5 b) 8 (52x) + 8(5x) = 6
-
Describe in detail how excessive praise might influence child development in negative ways. Tie this in with Erikson's ideas about Industry v Inferiority. How might excessive and unrealistic praise...
-
We start with some review problems A crate of mass 2 2 . 6 kg rests on a level surface, with a coefficient of kinetic friction 0 . 2 9 2 . You push on the crate with an applied force of 3 8 4 N ....
-
Linda Barnes has learned from prior studies that one out of five applicants gets admitted to top MBA programs in the country. She wishes to construct her own 90% confidence interval for the...
-
In July 2013, cnet.com listed the battery life (in hours) and luminous intensity (i. e., screen brightness, in cd/m2) for a sample of tablet computers. We want to know if screen brightness is...
-
What is the firm's cost of preferred stock? MINI CASE During the last few years, Harry Davis Industries has been too constrained by the high cost of capital to make many capital investments....
-
Harry Davis preferred stock is riskier to investors than its debt, yet the preferred's yield to investors is lower than the yield to maturity on the debt. Does this suggest that you have made a...
-
What are the two primary ways companies raise common equity? MINI CASE During the last few years, Harry Davis Industries has been too constrained by the high cost of capital to make many capital...
-
A light rail communter train has a top speed of 2 1 m / s . When stopping it slows at a rate of 1 . 6 5 m / s ^ 2 . ( a ) How long does it take the train to stop? ( b ) How much distance is needed?
-
Consider 3 positive charges q = 2 . 6 microcoulomb at the vertices of an equilateral triangle. Each side has length l = 1 . 7 cm . Find the magnitude of the total force ( in N ) on each of the...
-
A sealed treasure chest with a volume of 7 4 L and a mass of 1 4 9 kilograms is lying 1 7 meters below the ocean surface. What is the bouyant force on the chest ( in N ) ? The density of seawater is...

Study smarter with the SolutionInn App


