A solution is believed to contain one or more of the following ions: Cr 3+ , Zn
Question:
A solution is believed to contain one or more of the following ions: Cr3+, Zn2+, Fe3+, Ni2+. When the solution is treated with excess NaOH(aq), a precipitate forms. The solution in contact with the precipitate is colorless. The precipitate is dissolved in HCl(aq), and the resulting solution is treated with NH3(aq). No precipitation occurs. Based solely on these observations, what conclusions can you draw about the ions present in the original solution? That is, which ion(s) are likely present, which are most likely not present, and about which can we not be certain? Refer to Appendix D for solubility product and complex-ion formation data.
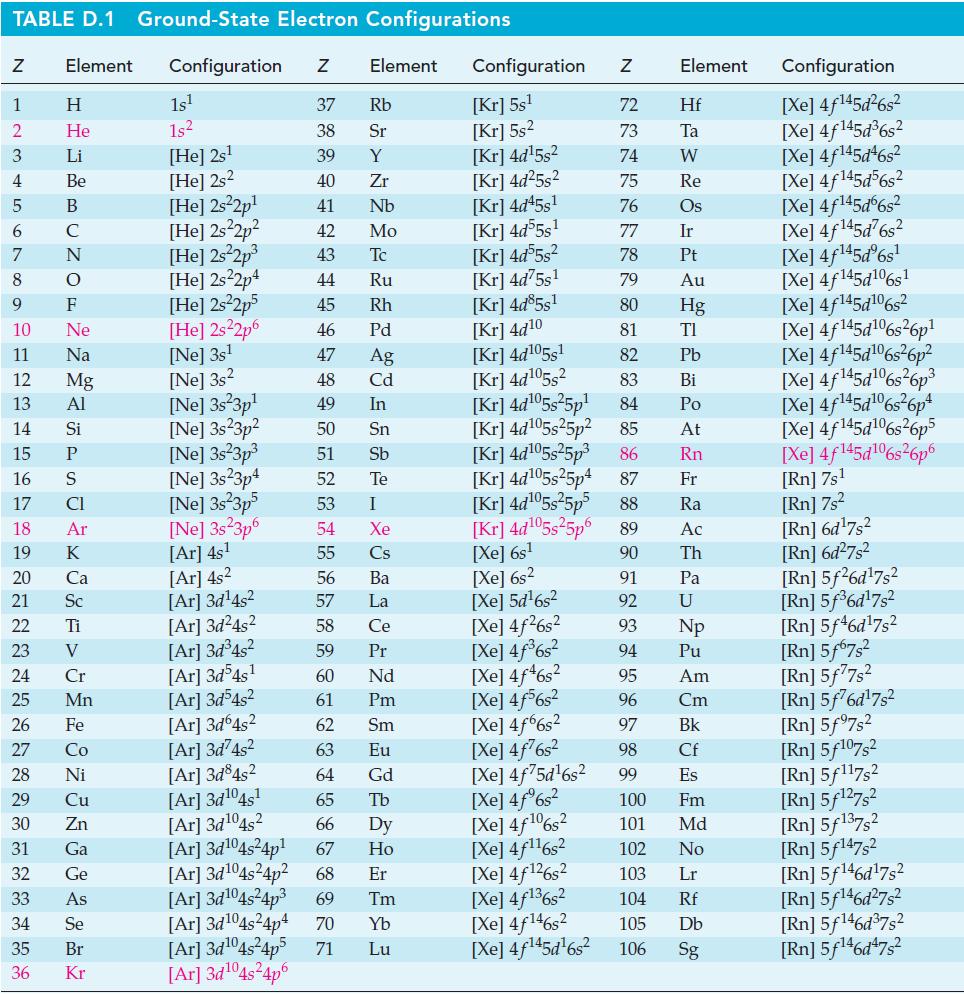
TABLE D.1 Ground-State Electron Configurations Element Configuration Z Z 1 2 3 4 6 7 8 9 5 B 10 11 12 13 14 15 16 17 18 19 20 25 27 29 HIGÅMUZONSUZ SE> 0 ≤ 2 3 2 3 5 3 3 4 8 5 2 30 Η 31 He 32 Li 33 Be C F Ne Na Mg Al 21 Sc 22 Ti 23 V 24 Si P CI Ar 26 Fe K Ca 28 Ni Cr Mn Co Cu Zn Ga Ge As 34 Se 35 Br 36 Kr 1s¹ 1s² [He] 2s¹ [He] 2s2 [He] 2s²2p¹ [He] 2s²2p² [He] 2s²2p³ [He] 2s22p4 [He] 2s²2p5 [He] 2s²2p6 [Ne] 3s¹ [Ne] 3s2 [Ne] 3s 3p¹ [Ne] 3s23p² 37 Rb 38 Sr 39 Y 40 Zr 41 Nb 42 Mo 43 Tc 44 Ru 45 Rh 46 Pd 47 Ag 48 Cd 49 In 50 Sn 51 Sb Te 52 53 I 54 Xe 55 Cs 56 Ba 57 La 58 Ce 59 Pr [Ar] 3d³4s² [Ar] 3d54s¹ 60 Nd 61 Pm 62 Sm 63 Eu [Ar] 3d³4s² [Ar]3d64s² [Ar]3d²4s² [Ar]3d845² [Ar] 3d¹04s¹ [Ar] 3d¹04s2 64 Gd 65 Tb 66 67 [Ar] 3d¹04s²4p¹ Dy Ho Er 69 Tm [Ar] 3d¹04s²4p² 68 [Ar]3d¹04s²4p³ [Ar] 3d¹04s²4p4 70 Yb Lu [Ne] 3s²3p³ [Ne] 3s23p4 [Ne] 3s 3p5 [Ne] 3s 3p6 [Ar] 4s¹ [Ar] 4s² [Ar]3d¹4s² [Ar]3d²4s² [Ar]3d¹04s²4p5 71 Element [Ar]3d¹04s²4p6 Configuration Z [Kr] 5s¹ [Kr] 5s² [Kr] 4d¹5s² [Kr] 4d²5s² [Kr] 4d45s¹ [Kr] 4d55s¹ [kr] 4d55s² [Kr] 4d75s¹ [Kr] 4d85s1 [Kr] 4d10 [Kr] 4d105s1 [Kr] 4d¹05s² [Kr] 4d¹05s²5p¹ [Kr] 4d¹05s25p² [Kr] 4d¹05s²5p³ [kr] 4d¹05s25p4 [Xe] 6s² [Xe] 5d¹6s² [Xe] 4f²6s² [Xe] 4f³6s² [Xe] 4f46s2 [Xe] 4f6s2 [Xe] 4f6s2 [Xe] 4f76s2 [Xe] 4f75d¹6s² [Xe] 4f%s2 [Xe] 4f106s2 [Xe] 4f¹¹6s² NRNKERKR [Xe] 4f126s2 [Xe] 4f136s2 [Xe] 4f146s2 [Xe] 4f¹45d¹6s² 72 Hf 73 Ta W 74 75 Re 76 Os 77 Element 78 79 80 81 82 83 84 85 86 87 Fr [Kr] 4d¹05s²5p5 88 Ra [Kr] 4d¹05s²5p6 89 Ac [Xe] 6s¹ 90 Th 91 92 93 94 95 96 97 98 99 Ir Pt Au Hg TI Pb Bi Po At Rn Pa U Np Pu Am Cm Bk Cf Es 100 Fm 101 Md 102 No 103 Lr 104 Rf 105 Db 106 Sg Configuration [Xe] 4f¹45d²6s² [Xe] 4f145d³6s² [Xe] 4f145d46s2 [Xe] 4f145d56s2 [Xe] 4f145d6s2 [Xe] 4f¹45d²6s² [Xe] 4f¹45dº6s¹ [Xe] 4f145d106s1 [Xe] 4f145d106s2 [Xe] 4f145d6s26p* [Xe] 4f145d106s36p? [Xe] 4f145d16s?6p3 [Xe] 4f145d6s®6p* [Xe] 4f145d16s26p5 [Xe] 4f145d106s26p6 [Rn] 7s¹ [Rn] 7s² [Rn] 6d¹7s² [Rn] 6d²7s² [Rn] 5f26d¹7s² [Rn] 5f³6d¹7s² [Rn] 5f46d¹7s2 [Rn] 5f67s² [Rn] 5f77s² [Rn] 5f76d¹7s² [Rn] 5f97s2 [Rn] 5f107,2 [Rn] 5f117s2 [Rn] 5f¹27s² [Rn] 5f137,2 [Rn] 5f147s2 Rn] 5f¹46d¹7s² [Rn] 5f¹46d²7s² [Rn] 5f¹46d³7s2 [Rn] 5f¹46d¹7s²
Step by Step Answer:

This question has not been answered yet.
You can Ask your question!

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Students also viewed these Sciences questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
THIRD AVENUE SOFTWARE HEALTH-CARE APP PROJECT This case is new for the ninth edition of Information Technology Project Management . The case provides an opportunity to apply agile and Scrum...
-
[A] Aon Corporations Mezzanine Preferred Stock In its 2002 annual report to shareholders, Aon Corporation described its mandatorily redeemable preferred stock as follows: In January 1997, Aon created...
-
Which of the following statements best describes the 'dilation' in rock mechanics? a. Movement of the rock along the shear direction during the direct shear tests b. Radial expansion of the intact...
-
Jason Company needs to estimate the inventory balance for its quarterly financial statements. The periodic inventory method is used. Records show that quarterly sales totaled $550,000, beginning...
-
Bentley Bros. Book Company publishes more than 250 fiction and nonfiction titles. Most of the companys books are written by southern authors and typically focus on subjects popular in the region. The...
-
Shiloh supplies equipment to the automotive and commercial vehicle markets and other industrial customers. It specializes in materials and designs that reduce vehicle weight and increase fuel...
-
The Metropolitan Bus Company (MBC) purchases diesel fuel from American Petroleum Supply. In addition to the fuel cost, American Petroleum Supply charges MBC $250 per order to cover the expenses of...
-
A house with a total living area of 2,500 square feet would cost $110 per square foot to reproduce new. It has an expected economic life of 50 years and is estimated to have an effective age of five...
-
A 0.960 g sample of impure hematite (Fe 2 O 3 ) is treated with 1.752 g of oxalic acid (H 2 C 2 O 4 2 H 2 O) in an acidic medium. Following this, the excess oxalic acid is titrated with 35.16 mL of...
-
Show that under the following conditions, Ba 2+ (aq) can be separated from Sr 2+ (aq) and Ca 2+ (aq) by precipitating BaCrO 4 (s) with the other ions remaining in solution: Use data from this and...
-
Italian Leather Goods Inc. began 2017 with an inventory of 50,000 units that cost $1,500,000. During the year, the store purchased merchandise on account as follows: March (40,000 units @ cost of...
-
Why do economists rely more on empirical evidence today than they did 100 years ago?
-
Antipoverty programs in the United States since the mid-1990s have focused on welfare-to-work programs that compel welfare recipients to take paid jobs. Some economists argue that these programs...
-
The total utility of your consumption of widgets is 40; it changes by 2 with each change in widgets consumed. The total utility of your consumption of wadgets is also 40 but changes by 3 with each...
-
You are buying your spouse, significant other, or close friend a ring. You decide to show your reasonableness and buy a cubic zirconium ring that sells at 150 the cost of a mined diamond and that any...
-
If the wage goes up 20 percent and the quantity of labor supplied increases by 5 percent, whats the elasticity of labor supply?
-
Anika and Jespar are married and have 2 children ages 16 and 14. Their adjusted gross income for the year is $98,000. What amount can they claim for the child credit? a. What amount can they claim...
-
Read the case study Richter: Information Technology at Hungarys Largest Pharma and answer the following question: How does the organization ensure the accuracy of the data it stores?
-
Delmont Company entered into these transactions during May 2012. 1. Purchased computers for office use for $30,000 from Dell on account. 2. Paid $4,000 cash for May rent on storage space. 3. Received...
-
During 2012, its first year of operations as a delivery service, Underwood Corp. entered into the following transactions. 1. Issued shares of common stock to investors in exchange for $100,000 in...
-
A tabular analysis of the transactions made during August 2012 by Nigel Company during its first month of operations is shown below. Each increase and decrease in stockholders?? equity is explained....
-
1. A bar made of steel (Young's modulus E = 200 GPa) is composed of three members joined together as illustrated in Figure 1. The bar is fixed at both left and right ends to rigid walls and subject...
-
Consider an isentropic flow in a convergent-divergent nozzle schematically shown in Figure Q2. The value of the temperature in the reservoir is 510 K, and the Mach number at the outlet section is...
-
A website has this code inside their login system. What could you type into the username box so that you could login without knowing the password? $user = $_POST['username']; $pass =...

Study smarter with the SolutionInn App


