(A) Use data from Table 7.2 to calculate the standard enthalpy of combustion of ethanol, C 2...
Question:
(A) Use data from Table 7.2 to calculate the standard enthalpy of combustion of ethanol, C2H5OH(l), at 298.15 K.
(B) Calculate the standard enthalpy of combustion at 298.15 K per mole of a gaseous fuel that contains C3H8 and C4H10 in the mole fractions 0.62 and 0.38, respectively.
Table 7.2
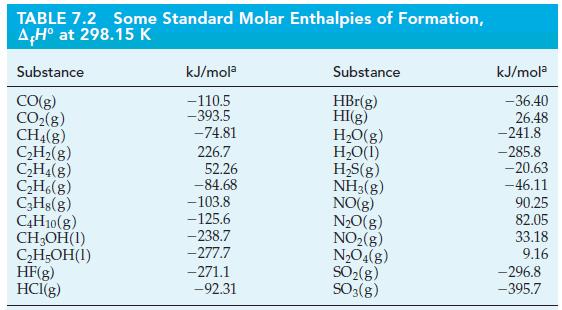
Transcribed Image Text:
TABLE 7.2 Some Standard Molar Enthalpies of Formation, A,Hº at 298.15 K Substance CO(g) CO₂(g) CH4(g) C₂H₂(g) C₂H4(8) C₂H6(g) C₂H8(g) C4H10(g) CH₂OH(1) C₂H5OH(1) HF(g) HCl(g) kJ/mola -110.5 -393.5 -74.81 226.7 52.26 -84.68 -103.8 -125.6 -238.7 -277.7 -271.1 -92.31 Substance HBr(g) HI(g) H₂O(g) H₂O(1) H₂S(g) NH3(g) NO(g) N₂O(g) NO₂(g) N₂O4(g) SO₂(g) SO3(g) kJ/mola - 36.40 26.48 -241.8 -285.8 -20.63 -46.11 90.25 82.05 33.18 9.16 -296.8 -395.7
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
1 Answer to Part A The balanced equation for the combustion of ethanol is C2H5OHl 3O2g 2CO2g 3H2O...View the full answer

Answered By

Morgan Njeri
Very Versatile especially in expressing Ideas in writings.
Passionate on my technical knowledge delivery.
Able to multitask and able to perform under pressure by handling multiple challenges that require time sensitive solution.
Writting articles and video editing.
Revise written materials to meet personal standards and satisfy clients demand.
Help Online Students with their course work.
4.90+
12+ Reviews
38+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Ethanol (C2H5OH) has been proposed as an alternative fuel. Calculate the standard enthalpy of combustion per gram of liquid ethanol.
-
The standard enthalpy of combustion of solid urea (CO (NH2)2) is -632 kl mol-1 at 298 K and its standard molar entropy is 104.60 J K-1 mol-1, Calculate the standard Gibbs energy of formation of urea...
-
The molar heat capacity of ethane is represented in the temperature range 298 K to 400 K by the empirical expression Cp,m/ (J K-1 mol-1) = 14.73 + 0.1272(T/K). The corresponding expressions for C(s)...
-
What is the wavelength of light if its frequency is 1.009 106 Hz?
-
1. Which two short-term liquidity ratios measure how frequently a company collects its accounts? 2. What measure reflects the difference between current assets and current liabilities? 3. Which two...
-
MRP II stands for: a) Material resource planning. b) Management requirements planning. c) Management resource planning. d) Material revenue planning. e) Material risk planning.
-
Describe the V-Model, its characteristics, and shortcomings.
-
With regard to accounting for private, not-for-profit health care entities, do the following: a. Outline the accounting required, under FASB guidance, for a(n): (1) Endowment gift received in cash....
-
Now, it's time to buy that sweet new ride. If you are going to live in the mountains of North Carolina, there is an unwritten code that you must own a Subaru. So, you are going to buy a brand new...
-
A factory has five assembly lines. Each assembly line is down (not working) p percent of the time. What must p equal so that there is a 95 percent chance that at least one assembly line is working?
-
(A) The overall reaction that occurs in photosynthesis in plants is Determine the standard enthalpy of formation of glucose, C 6 H 12 O 6 (s), at 298.15 K. (B) A handbook lists the standard enthalpy...
-
(A) The standard enthalpy of formation for the amino acid leucine, C 6 H 13 O 2 N(s), is -637.3 kJ/mol. Write the chemical equation to which this value applies. (B) How is r H for the following...
-
What is merchant banking?
-
Assuming Jeff has a mild chronic disease, and he has a private hospital health insurance. His salary before tax is $170,000. And assuming he has sold $30,000 of mutual fund units after he bought them...
-
Describe the classic steps for identifying an endocrine gland. Are these steps practical for identifying the sources of all the hormones we know of today? Explain.
-
Your portfolio which consists of 46% of stock J, and 54% of stock K. J has daily Standard Deviation 3% while K has daily Standard Deviation 4%. The correlation of two stocks is -0.6. What is the 1...
-
Add as indicated. Write all answers in lowest terms. 3 - + 2 X X-7 3X 2 = X-7 (Simplify your answer.)
-
f(x)=5x+2 g(x)=3x-7 (f-g)(-2): (F) (2)
-
Study the Excel regression output that follows. How many predictors are there? What is the equation of the regression model? Using the key statistics discussed in this chapter, discuss the strength...
-
When an electric field is applied to a shallow bath of vegetable oil, why do tiny bits of thread floating in the oil align with the field like compasses in a magnetic field?
-
Ratio Computations and Effect of Transactions Presented below is information related to Leland Inc. (a) Compute the following ratios or relationships of Leland Inc. Assume that the ending account...
-
Current Liability Entries and Adjustments Described below are certain transactions of Edward son Corporation. The company uses the periodic inventory system. 1. On February 2, the corporation...
-
Liability Entries and Adjustments Listed below are selected transactions of Schultz Department Store for the current year ending December 31. 1. On December 5, the store received $500 from the...
-
(1) Write the equation of the circle with center (3, 4) and radius 2. (2) Suppose P is a point on circle C with radial line y =3x-2. What is the slope of the line tangent to C at P? (3) What is the...
-
What role does symbolism and storytelling play in the reinforcement of organizational culture, and how can leaders effectively leverage these elements to strengthen cultural identity ?
-
How does organizational culture shape the perception and acceptance of diversity, equity, and inclusion (DEI) initiatives, and what are the key factors that drive a successful DEI transformation ?

Study smarter with the SolutionInn App


