(A) What is the value of K for the dissociation of HF(g) into its elements at 298...
Question:
(A) What is the value of K for the dissociation of HF(g) into its elements at 298 K? Use data from Table 22.6.
(B) Use data from Table 22.6 to determine K and the percent dissociation of HCl(g) into its elements at 298 K.
Table 22.6
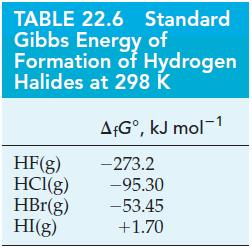
Transcribed Image Text:
TABLE 22.6 Standard Gibbs Energy of Formation of Hydrogen Halides at 298 K AfGº, kJ mol-1 -273.2 HF(g) HCl(g) -95.30 HBr(g) -53.45 HI(g) +1.70
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
A Calculating the value of K for the dissociation of HFg into its elements at 298 K Equation 2HFg H2...View the full answer

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
What is the value of K for the dissociation of HI(g) into its elements at 298 K?
-
(A) Use data from Example 15-2 to determine the value of K at 298 K for the reaction (B) For the reaction NO(g) + 1/2 O 2 (g) NO 2 (g) at 184 C, K = 1.2 x 10 2 . What is the value of K at 184 C for...
-
Simba and Disney, LP (a partnership) each own 1,500 shares in Lion King, Inc. Disney, LP is owned 1/3 by Simba. Simba and Disney have owned the shares in Lion King Inc. for 15 years. Simba, wanting...
-
In a country with a fixed exchange rate system the rise of inflation will result in: O Home currency depreciation Currency appreciation in real terms Floating of home currency O Inflow of foreign...
-
What danger is there in basing a managers bonus on reported net income?
-
If regulations restricting institutional investors to investment grade bonds were lifted, what do you think would happen to the spreads between yields on investment grade and speculative grade bonds?
-
Assuming the same data as given in problem 9, was the well in each case profitable? Discuss your answer. Problem 9:- Property cost (acquisition cost). Drilling cost (one well). Estimated completion...
-
In your audit of Canyon Outdoor Provision Company's financial statements, the following transactions came to your attention: 1. Canyon Outdoor's operating lease for its main store is with York...
-
x Determine the solution of the following system of equations via row reduction of the corresponding augmented matrix: x+5y+3z 11 y 23 z = 4 = 11 y + 21 z = 1 Give your answer in the form of a row...
-
Fluorine is able to stabilize elements in very high oxidation states. For each of the elements Na, Mg, Al, Si, P, S, and Cl, give the formula of the highest oxidationstate of fluoride that is known...
-
Do you expect the ions ICl 2 + and ICl 2 to have the same shape? Explain.
-
Arctic explorers are unsure if they can use a 5-kW motor-driven heat pump to stay warm. It should keep their shelter at 15C. The shelter loses energy at a rate of 0.5 kW per degree difference to the...
-
The Taylor rule is a simple equation that describes movements in the target federal funds rate. It suggests that: a. When inflation rises, the FOMC raises the target rate by 1 times the increase in...
-
Show the impact on the Federal Reserves balance sheet of a foreign exchange market intervention where the Fed purchases $5,000 worth of foreign exchange reserves. Explain what impact, if any, the...
-
One goal of the regulatory reforms that followed the 2007-2009 financial crisis was to address the too-big-to-fail problem associated with large institutions. How did the reforms try to address this...
-
In 2012, the Federal Reserve joined many other central banks by making explicit a numerical target for inflation. Explain how stating that an annual inflation rate of 2 percent over the long run is...
-
A euro-area country that runs very large public deficits or shows a persistently high and rising debt-to-GDP ratio violates the provisions of a 2012 treaty aimed at promoting fiscal stability....
-
Kuerten Manufacturers sues the Rafter Corporation for patent infringement. The court upholds Kuertens claim and requires Rafter to pay Kuerten $2,000,000 in damages. However, the court does not allow...
-
Refer to the table to answer the following questions. Year Nominal GDP (in billions) Total Federal Spending (in billions) Real GDP (in billions) Real Federal Spending (in billions) 2000 9,817 578...
-
Information about Sunburst is presented in E6-4. Additional data regarding the company's sales of Xpert snowboards are provided below. Assume that Sunburst uses a perpetual inventory system....
-
Brooks Hardware reported cost of goods sold as follows. Brooks made two errors:1. 2011 ending inventory was overstated by $2,000.2. 2012 ending inventory was understated by $5,000.InstructionsCompute...
-
Sprague Company reported these income statement data for a 2-year period. Sprague Company uses a periodic inventory system. The inventories at January 1, 2011, and December 31, 2012, are correct....
-
ALTERNATIVE FIXED COST VARIABLE COST Buy $200,000 per year $15 per unit Make $50,000 per year $20 per unit What does the company save for the year by selecting the low-cost option at an annual...
-
Violet Corp. reported net income in the current year of $120,000. The following transactions were recorded in the current year: Cash dividends of $23,000 were paid to shareholders. Stock dividends...
-
How do authors employ allusions as tools of satire and parody, subverting established norms, tropes, and conventions by appropriating and recontextualizing familiar cultural references to critique...

Study smarter with the SolutionInn App


