Acetyl acetone undergoes an isomerization to form a type of alcohol called an enol. The enol, abbreviated
Question:
Acetyl acetone undergoes an isomerization to form a type of alcohol called an enol.
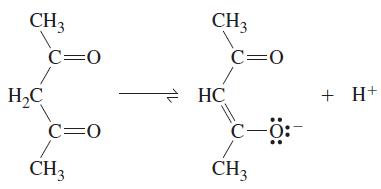
The enol, abbreviated acacH, can act as a bidentate ligand as the anion acac–. Which of the following compounds are optically active: Co(acac)3; trans-[Co(acac)2(H2O)2]Cl2; cis-[Co(acac)2(H2O)2]Cl2?
Transcribed Image Text:
CH3 H2C c=0 c=0 CH3 CH3 HC c=0 с-ӧ: CH3 + H+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
The acetylacetone molecule acacH can undergo isomerization to form the enol form ...View the full answer

Answered By

Omar ELmoursi
I'm Omar, I have Bachelor degree in Business and Finance, My unique approach is to help students with questions and assignments, I can teach Business, Math, Accounting, Managerial Accounting, Economy, Human resources management, organizational behavior, project management, I have experience dealing with different types of students and teach them how to deal with different types of exercises.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family. The Incisors own a rental beach house in Hawaii. The beach house was rented for the full year during 2012...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Technology World tsad the following revenue and expenses during the month ended July 31, Fees for computer repairs Advertising expense Salaries expense Telephone expenses fees for printer repairs...
-
Letha Enterprises purchased a new van on January 1, 2011, for $35,000. The estimated life of the van was four years or 76,000 miles, and its salvage value was estimated to be $3,000. Compute the...
-
How do environmental factors affect response rates in personal interviews? How can we overcome these environmental problems?
-
Consider the following cash flow profile and assume MARR is 10 percent/year. a. What does Descartes' rule of signs tell us about the IRR(s) of this project? b. What does Norstrom's criterion tell us...
-
Woodruff Manufacturing produces a subassembly used in the production of jet aircraft-engines. The assembly is sold to engine manufacturers and aircraft maintenance facilities. Projected sales for the...
-
Direct labor-hours Machine-hours Total fixed manufacturing overhead cost Variable manufacturing overhead per machine-hour Variable manufacturing overhead per direct labor-hour Required: 1. Compute...
-
A tabulation of formation constants lists the following log 10 K values for the formation of [Cu(NH 3 ) 4 ] 2+ : log 10 K 1 = 4.28, log 10 K 2 = 3.59, log 10 K 3 = 3.00, and log 10 K 4 = 2.18....
-
Provide a valence bond description of the bonding in the Cr(NH 3 ) 6 3+ ion. According to the valence bond description, how many unpaired electrons are there in the Cr(NH 3 ) 6 3+ complex? How does...
-
The scores of all applicants taking an aptitude test required by a law school have a normal distribution with a mean of 420 and a standard deviation of 100. A random sample of 25 scores is taken. a....
-
An elevator bank monitoring system shows the clock time in hours and minutes on multiple displays. The time is generated by a programmable 16 - bit timer, which uses a 50 - kHz clock signal for...
-
Harmon, the owner of a suburban house, rented it for one year to Wagner and his wife, who had just moved into the city from another state. Two months after moving into the house, the Wagners divorced...
-
An intruder entered through a window and raped McCutchen in her apartment. McCutchen sued the landlord, Ten Associates, for failure to provide adequate security and failure to warn her of the risk of...
-
Suppose x is a 16 - bit BAM word representing the angle 225 , and y is another 16 - bit BAM word representing 157.5 . Using binary arithmetic, show that x + y = 22.5.
-
Knight purchased an apartment building that was occupied by tenants Hallsthammer, Decaprio, and Breit. The day after acquiring the building, Knight informed the tenants that their rent was being...
-
Tina owns and operates Timely Turn Tables (TTT) as a sole proprietorship. TTTs taxable income during the current year is $80,000. In addition to the TTT income, Tina has the following income and...
-
A crop-dusting plane flies over a level field at a height of 25 ft. If the dust leaves the plane through a 30 angle and hits the ground after the plane travels 75 ft, how wide a strip is dusted? See...
-
Orosco Supply Co. has the following transactions related to notes receivable during the last 2 months of 2010. Nov. 1 Loaned $15,000 cash to Sally Givens on a 1-year, 10% note. Dec. 11 Sold goods to...
-
Record the following transactions for Sandwich Co. in the general journal. 2010 May 1 Received a $7,500, 1-year, 10% note in exchange for Julia Gonzalezs outstanding accounts receivable. Dec. 31...
-
Singletary Company had the following select transactions. Apr. 1, 2010 Accepted Wilson Companys 1-year, 12% note in settlement of a $20,000 account receivable. July 1, 2010 Loaned $25,000 cash to...
-
Find the energy dissipated during a cycle of simple harmonic motion given by x(t) = 0.2 sin(wat) m by a viscously damped single-degree-of-freedom system with the following parameters: m = 10 kg, c =...
-
Bond 1- Term to maturity is 3 years, Face value is 100, coupon rate is 2.5%, yield is 3.54%, and portfolio holding is 175 million; Bond 2: Term to maturity is 10 years, Face value is 100, coupon rate...
-
An aluminium hollow box column of square cross section is fixed at the base and free at the top. The width of each side is b = 150 mm and the thickness t = 10 mm. A compressive load P = 160 KN acts...

Study smarter with the SolutionInn App


