Assign formal charges to each of the atoms in the following structures. (a) (b) (c) S N
Question:
Assign formal charges to each of the atoms in the following structures.
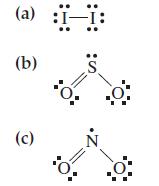
Transcribed Image Text:
(a) (b) (c) S N
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
To assign formal charges to the atoms in the structures well follow these steps Count the valence electrons for each atom in its ground state This is ...View the full answer

Answered By

Larlyu mosoti
I am a professional writer willing to do several tasks free from plagiarism, grammatical errors and submit them in time. I love to do academic writing and client satisfaction is my priority. I am skilled in writing formats APA, MLA, Chicago, and Harvard I am a statistics scientist and I can help out in analyzing your data. I am okay with SPSS, EVIEWS, MS excel, and STATA data analyzing tools.
Statistical techniques: I can do linear regression, time series analysis, logistic regression, and some basic statistical calculations like probability distributions. . I'm ready for your working projects!
Services I would offer:
• Academic writing.
• Article writing.
• Data entry.
• PDF conversion.
• Word conversion
• Proofreading.
• Rewriting.
• Data analyzing.
The best reason to hire me:
- Professional and Unique work in writing.
- 100% satisfaction Guaranteed
- within required time Express delivery
- My work is plagiarism Free
- Great communication
My passion is to write vibrantly with dedication. I am loyal and confident to give my support to every client. Because Client satisfaction is much more important to me than the payment amount. A healthy client-contractor relationship benefits in the longer term. Simply inbox me if you want clean work.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Below are the equations for four impulse responses. The plot below shows four step responses. Using the Laplace convolution technique, which impulse response goes with which step response (A, B, C,...
-
Write Lewis structures that obey the octet rule for each of the following, and assign oxidation numbers and formal charges to each atom: (a) OCS, (b) SOCl2 (S is bonded to the two Cl atoms and to the...
-
Write Lewis structures for each molecule or ion. Include resonance structures if necessary and assign formal charges to all atoms. If necessary, expand the octet on the central atom to lower formal...
-
It is desired to control the exit temperature T2 of the beat exchanger shown In Figure by adjusting the steam flow rate w3, unmeasured disturbances occur in inlet temperature T1. The dynamic behavior...
-
Assume that a company chooses an accelerated method of calculating depreciation expense for financial statement reporting purposes for an asset with a five-year life. Required: State the effect...
-
What are the capabilities of supply chain management software? How do ERP and SCM relate?
-
Sarah Lou Bakery, Inc., reported a prior-period adjustment in 2008. An accounting error caused net income of prior years to be overstated by \(\$ 5,000\). Retained earnings at December 31, 2007, as...
-
Delille Company manufactures both traditional toothpaste and gel toothpaste, with each type of toothpaste produced in separate departments. Three support departments support the production...
-
The Megan Companies (MC) IPO offer price was $15 (per share). When MC shares began trading in the public market, the price was $13.50. The MC IPO was: a.) Underpriced by 10% b.) Overpriced by 11.11%...
-
Both oxidation state and formal charge involve conventions for assigning valence electrons to bonded atoms in compounds, but clearly they are not the same. Describe several ways in which these...
-
Assign formal charges to each of the atoms in the following structures. (a) [H-C=C:] (b) :0: C :0: 72- O: (c) [CH3-CH-CH3]+
-
Consider the reaction: If a reaction mixture initially contains 0.110 M CO and 0.110 M H 2 O, what will the equilibrium concentration of each of the reactants and products be? CO(g) + HO(g) = CO(g) +...
-
The following is a fictitious list of figures for a given year in billions of dollars. Exports Capital Consumption Allowance 1561 1215 Corporate Income Taxes 871 Wages 6333 Government expenditure...
-
Taylor (70kg) was standing still on frictionless ice. He threw a 5.0kg rock straight east at 11m/s. How fast was Taylor moving immediately after the rock left his hand? Please explain and show all...
-
Write a thesis on income equality in India, write something about Income Equality in India, and compare it with Income Equality in Canada.
-
For your final project, you'll need access to the case file from Harvar and the link for purchase is https://hbsp.harvard.edu/import/1046501Links to an external site. (Links to an external site.)(...
-
Yaster Gadgets manufactures and sells a smartphones per week. The weekly price-demand and cost equations are, respectively, p = 484 -0.45 x and C(x) = 20,081 +20x. Suppose Yaster Gadgets wants to...
-
Evaluate how well Frank and Joe have handled the problem. Evaluate Larson Cements internal control processes as they apply to this case. Joe Frecaso was recently hired as the controller for Larson...
-
At the beginning of its fiscal year, Lakeside Inc. leased office space to LTT Corporation under a seven-year operating lease agreement. The contract calls for quarterly rent payments of $25,000 each....
-
What is a master budget? Briefly describe its contents.
-
Why is the sales forecast the starting point in budgeting?
-
As a practical matter, planning and control mean exactly the same thing. Do you agree? Explain.
-
During a domestic quarrel a wife continually taunted her husband by degrading him and telling him he was a "lousy lover" and that she wanted to be rid of him. When the husband tried to persuade her...
-
Mr. Andrews (age 75) taught poetry in the Cook County school district. He is recently widowed, his wife having passed away a year ago after a lengthy illness; she had breast cancer for the last five...
-
How does the blurring of boundaries between work and personal life in a remote work environment impact employee stress levels, productivity, absenteeism, turnover rates, and organizational stability...

Study smarter with the SolutionInn App


