Calculate the vapor pressure of water at 35.0 C using data from Tables 12.4 and 12.5. Tables
Question:
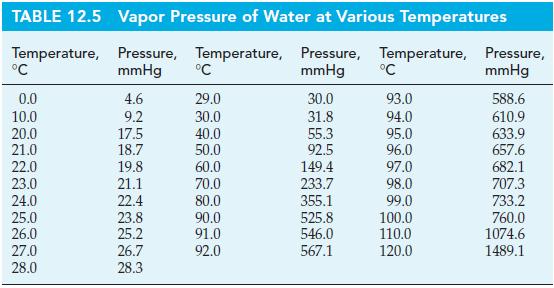
Calculate the vapor pressure of water at 35.0 °C using data from Tables 12.4 and 12.5.
Tables 12.4
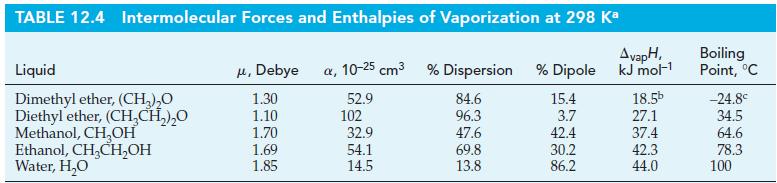
Tables 12.5
Transcribed Image Text:
TABLE 12.4 Intermolecular Forces and Enthalpies of Vaporization at 298 Ka Liquid Dimethyl ether, (CH₂)₂0 Diethyl ether, (CH₂CH₂)₂0 Methanol, CH₂OH Ethanol, CH₂CH₂OH Water, H₂O μ, Debye a, 10-25 cm³ % Dispersion % Dipole 52.9 84.6 96.3 47.6 69.8 13.8 1.30 1.10 1.70 1.69 1.85 102 32.9 54.1 14.5 15.4 3.7 42.4 30.2 86.2 AvapH, kJ mol-¹ 18.5b 27.1 37.4 42.3 44.0 Boiling Point, °C -24.8° 34.5 64.6 78.3 100
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
Analyze Starting with the ClausiusClapeyron equation we recognize that we need four pieces of data t...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The vapor pressure of water at various temperatures follows: (a) Draw a scatter diagram of these data. What type of relationship seems appropriate in relating y to x? (b) Fit a simple linear...
-
In the process of distillation, a mixture of two (or more) volatile liquids is first heated to convert the volatile materials to the vapor state. Then the vapor is condensed, reforming the liquid....
-
Suppose you were in charge of establishing economic policy for a small island country whose only industry is trapping. When people aren't trapping, they're enjoying leisure time with their families....
-
Suppose that a 7% semi-annual coupon bond with a time to maturity of 8 years and a par value of $100 has a price of $106,4. This bond is first callable in 6 years at a redemption price of $104,8....
-
In April, F. W. Lang Company purchased an ice cream freezer and refrigeration compressor unit from Fleet for $2,160. Although the parties agreed to a written installment contract providing for an...
-
How is blockchain technology being used to reduce poverty and help clean up our oceans?
-
Give a brief outline of ways that Sales Manager Harold Horne can contribute to the marketing planning process at Auckland Engineering.
-
On July 1, 2010, Spahn Co. pays $18,000 to Randle Insurance Co. for a 3-year insurance contract. Both companies have fiscal years ending December 31. For Spahn Co., journalize and post the entry on...
-
Isabella is a 14-year-old Hispanic bisexual female who has come into Department of Child Safety (DCS) care due to neglect. Isabella's mother Martina is 35 years old, a single mother, has an upcoming...
-
Recall the discussion of dew and frost formation. Do the surroundings absorb or lose heat when water vapor condenses to dew or frost? Is the quantity of heat per gram of H 2 O(g) condensed the same...
-
One of the following substances is a liquid at room temperature and the others are gaseous: CH 3 OH; C 3 H 8 ; N 2 ; N 2 O. Which do you think is the liquid? Explain.
-
The Hermite functions (Sect. 4.3.3) are a basis for the space \(\Omega=\) \(C_{R^{1}}^{\infty} \cap L_{R^{1}}^{2}\) of functions defined on the unbounded domain \(R^{1}\). Determine the coordinates...
-
Esquire Incorporated uses the LIFO method to report its inventory. Inventory at the beginning of the year was $ 7 0 0 , 0 0 0 ( 2 8 , 0 0 0 units at $ 2 5 each ) . During the year, 9 6 , 0 0 0 units...
-
2. The transactions of Spade Company appear below: 1/1 K. Spade, owner, invested $100,750 cash in the company in exchange for common stock 1/5 The company purchased supplies for $1,250 cash. 1/10 The...
-
Clonex Labs, Incorporated, uses the weighted - average method in its process costing system. The following data are available for one department for October: Units Percent Completed Materials...
-
Hydro Sports budgets overhead cost of $793,500 for the year; of this amount, $586,500 is traceable to the Assembly department and $207,000 is traceable to the Finishing department. The company...
-
Curtis Corporation's contribution margin is $25 per unit for Product A and $30 for Product B. Product A requires 2 machine hours and Product B requires 4 machine hours. How much is the contribution...
-
Unilever sells products such as Ben & Jerrys and Breyers ice cream, Hellmans mayonnaise, and Lipton tea. The company published a 33-page, printed version of its 2008 sustainability report overview....
-
U.S. households have become smaller over the years. The following table from the 2010 GSS contains information on the number of people currently aged 18 years or older living in a respondent's...
-
Calculating Project NPV you have been hired as a consultant for Pristine Urban-Tech Zither, Inc. (PUTZ), manufacturers of fine zithers. The market for, zithers is growing quickly. The company bought...
-
Project Evaluation Aguilera Acoustics (AAI), Inc. projects unit sales for a new seven-octave voice emulation implant as follows: Production of the implants will require $1,500,000 in net working...
-
Calculating Required Savings a proposed cost-saving device has an installed cost of $540,000. The device will be used in a five-year project but is classified as three-year MACRS property for tax...
-
You have been hired as the new controller for the Ralston Company. Shortly after Joining the company In 2 0 2 4 , you discover the following errors related to the 2 0 2 2 and 2 0 2 3 financlal...
-
Harris Company manufactures and sells a single product. A partially completed schedule of the company s total costs and costs per unit over the relevant range of 62, 0 0 0 to 1 0 8 , 0 0 0 units is...
-
The T . L . Lin Company manufactures a variety of natural fabrics for the clothing industry in a suburb of Shanghai. The following data in Chinese currency called yuan ( CNY ) pertain to the month of...

Study smarter with the SolutionInn App


