Determine the values of K p from the K c values given. (a) NO4(g) K (b) 2
Question:
Determine the values of Kp from the Kc values given.
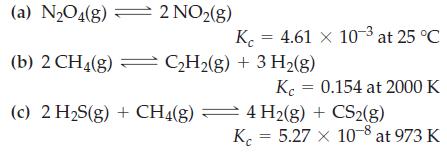
Transcribed Image Text:
(a) N₂O4(g) K (b) 2 CH4(g) = C₂H₂(g) + 3 H₂(g) (c) 2 H₂S(g) + CH4(g) 2 NO₂(g) = 4.61 x 10-3 at 25 °C Kc = 0.154 at 2000 K 4 H2(g) + CS2(g) K = 5.27 x 10-8 at 973 K
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
Here are the conversion factors between Kp and Kc for the given reactions Reaction Kp Kc Conversion ...View the full answer

Answered By

Arun kumar
made more than four thousand assignments
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
You have been assigned the task of measuring the equilibrium constant for the reaction N 2 O 4 2NO 2 as a function of temperature. To do so, you evacuate a rigid 2-liter vessel equipped with a...
-
Consider the nanofluid of Example 2.2. Example 2.2 (a) Calculate the Prandtl numbers of the base fluid and nanofluid, using information provided in the example problem. (b) For a geometry of fixed...
-
A 0.831-g sample of SO3 is placed in a 1.00-L container and heated to 1100 K. The SO3 decomposes to SO2 and O2: At equilibrium the total pressure in the container is 1.300 atm. Find the values of Kp...
-
Because Natalie has been so successful operating Cookie Creations, Katy would like to have Natalie become her partner. Katy believes that together they will create a thriving cookie-making business....
-
Ogden Corporation has compiled the following information on a capital expenditure proposal: (1) The projected cash inflows are normally distributed with a mean of $36,000 and a standard deviation of...
-
The adjusted trial balance of Mendoza Company shows the following data pertaining to sales at the end of its fiscal year October 31, 2014: Sales Revenue $820,000, Freight Out $16,000, Sales Returns...
-
The bookkeeper of Beautiful World Landscaping, Inc., prepared the companys balance sheet while the accountant was ill. The balance sheet contains numerous errors. In particular, the bookkeeper knew...
-
Clothes, Inc., has an average annual demand for red, medium polo shirts of 25,000 units. The cost of placing an order is $80 and the cost of carrying one unit in inventory for one year is $25....
-
4. The World Energy Council estimate for Canadian bitumen is 1.7 trillion barrels of BIP (2/3 of world bitumen resource), if that was counted in global reserves, how would that change the oil...
-
(A) Equilibrium is established in a 3.00 L flask at 1405 K for the reaction 2 H 2 S(g) 2 H 2 (g) + S 2 (g). At equilibrium, there is 0.11 mol S 2 (g), 0.22 mol H 2 (g), and 2.78 mol H 2 S(g). What...
-
(A) The reaction N 2 O 4 (g) 2 NO 2 (g) has r H = +57.2 kJ mol -1 . Will the amount of NO 2 (g) formed from N 2 O 4 (g) be greater at high or low temperatures? (B) The enthalpy of formation of NH 3...
-
With respect to ethics, what are the responsibilities of the Public Company Accounting Over sight Board? What is the source of the Board's authority?
-
Suppose a bank currently has $240,000 in deposits and $23,000 in reserves. The required reserve ratio is 13%. If at the end of the day, there is an unexpected withdrawal of $4,000 in reserves, what...
-
The growth rate for the firm's common stock is 7%. The firm's preferred stock is paying an annual dividend of $5. What is the preferred stock price if the required rate of return is 8%?
-
1) Calculate the magnitude of the electric field generated (8.4x10^0) m from an electron, giving your answer in N/C to 2 significant figures. Magnitudes must be positive. 2) A non-conducting...
-
What role does the span of control play in shaping managerial effectiveness, and how does it relate to organizational hierarchy and employee autonomy ?
-
The last dividend paid by Klein, Inc. was $2.20. Klein's growth rate is expected to be 10 percent for two years, after which dividends are expected to grow at a rate of 6 percent forever. The...
-
Compare screening decisions with preference decisions.
-
The following selected information was taken from Sun Valley Citys general fund statement of revenues, expenditures, and changes in fund balance for the year ended December 31, 2019: Revenues:...
-
Alcoa Inc. is the worlds largest producer of aluminum products. One product that Alcoa manufactures is aluminum sheet products for the aerospace industry. The entire output of the Smelting Department...
-
Domino Foods, Inc., manufactures a sugar product by a continuous process, involving three production departmentsRefining, Sifting, and Packing. Assume that records indicate that direct materials,...
-
The chief cost accountant for Mountain Glade Beverage Co. estimated that total factory overhead cost for the Blending Department for the coming fiscal year beginning March 1 would be $546,000, and...
-
Accounting Education Ltd provides tutoring services for students who plan to complete the Association of Chartered Certified Accountants program. Accounting Education Ltd has two operating divisions:...
-
Carla Vista, Inc. uses a flexible budget for manufacturing overhead based on machine hours. Variable manufacturing overhead costs per machine hour are as follows: Indirect labor Indirect materials...
-
Solera Company manufactures a standard product SV1 which is a smart vacuum cleaner. SV1 currently fully utilises the available production capacity of 6,000 machine hours per period. Solera Company...
PHP Application Development With NetBeans Beginners Guide 1st Edition - ISBN: 1849515808 - Free Book

Study smarter with the SolutionInn App


