Estimate the percent dissociation of Cl 2 (g) into Cl(g) at 1 atm total pressure and 1000
Question:
Estimate the percent dissociation of Cl2(g) into Cl(g) at 1 atm total pressure and 1000 K. Use data from Appendix D and equations found elsewhere in this text, as necessary.
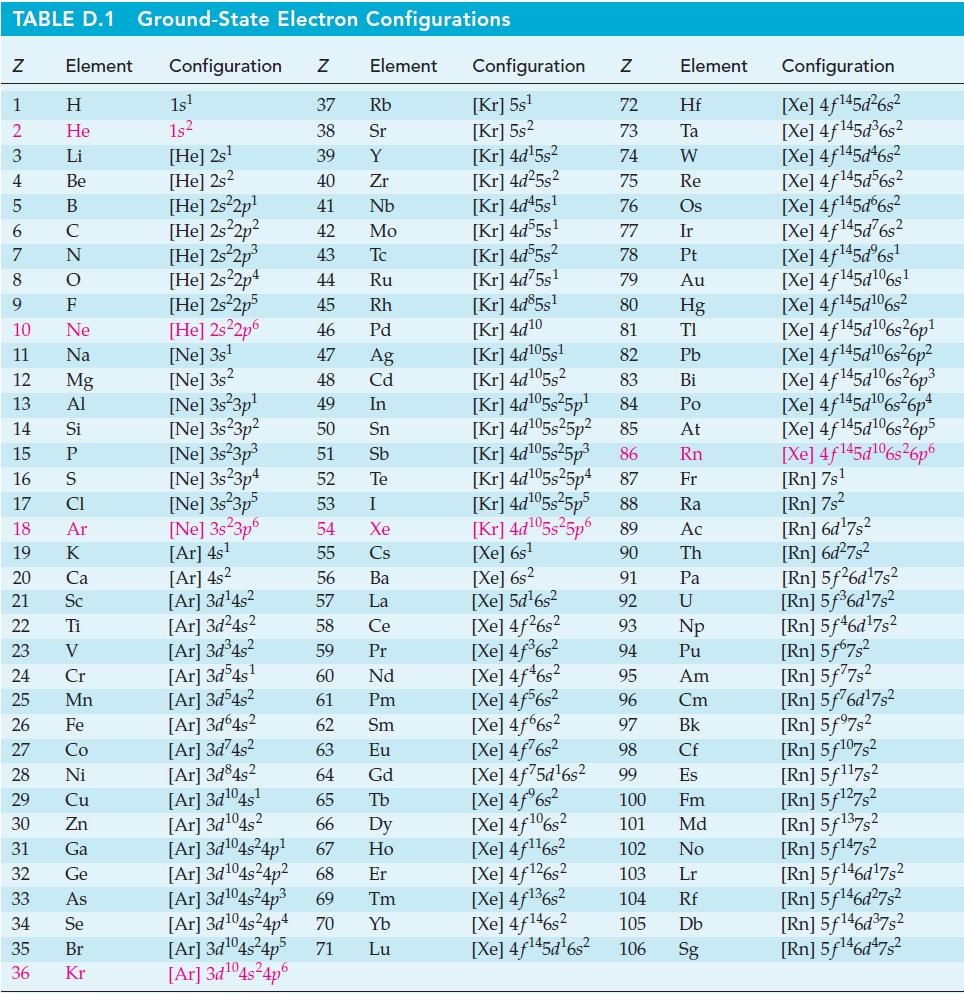
Transcribed Image Text:
TABLE D.1 Ground-State Electron Configurations Element Configuration Z Z 1 2 3 4 5 6 7 8 9 HIG&LUZONSUZ SE> 0 ≤ 2 8 2 3 5 3 3 2 2 5 2 He 10 11 12 13 14 15 16 17 18 19 20 21 Sc 22 23 24 25 Mn Mg 26 27 28 Ni 29 30 31 32 33 34 35 36 Zn Ga Ge 1s¹ 1s² [He] 2s¹ [He] 2s2 [He] 2s²2p¹ [He] 2s²2p² [He] 2s²2p³ [He] 2s22p4 [He] 2s²2p5 [He] 2s²2p6 [Ne] 3s¹ [Ne] 3s2 [Ne] 3s 3p¹ [Ne] 3s 3p² 37 Rb 38 Sr 39 Y 40 Zr 41 Nb 42 Mo 43 Tc 44 Ru 45 Rh 46 Pd 47 Ag 48 Cd 49 In 50 Sn 51 Sb 52 Te 53 54 55 56 57 58 59 60 61 62 [Ar]3d²4s² 63 [Ar] 3d845² 64 66 [Ar]3d¹04s¹ 65 [Ar]3d¹04s2 [Ar]3d¹04s²4p¹ [Ar] 3d¹04s²4p² 68 67 [Ar] 3d¹04s²4p³ [Ne] 3s²3p³ [Ne] 3s23p4 [Ne] 3s²3p5 [Ne] 3s 3p6 [Ar] 4s¹ [Ar] 4s² [Ar]3d¹4s² [Ar]3d²4s² [Ar] 3d³4s² [Ar]3d54s¹ [Ar]3d³4s² [Ar] 3d64s² Element [Ar]3d¹04s²4p4 70 [Ar]3d¹04s²4p5 71 [Ar]3d¹04s²4p6 I Xe Cs Ba La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er 69 Tm Yb Lu Configuration Z [Kr] 5s¹ [Kr] 5s² [Kr] 4d¹5s² [Kr] 4d²5s² [Kr] 4d45s¹ [kr] 4d55s¹ [Kr] 4d55s² [Kr] 4d75s¹ [Kr] 4d85s1 [Kr] 4d10 [Kr] 4d105s1 [Kr] 4d¹05s² [Kr] 4d¹05s²5p¹ [Kr] 4d¹05s25p² [kr] 4d¹05s²5p³ [kr] 4d¹05s25p4 [Xe] 6s² [Xe] 5d¹6s² [Xe] 4f²6s² [Xe] 4f³6s² [Xe] 4f46s2 [Xe] 4f6s2 [Xe] 4f6s2 [Xe] 4f²6s² [Xe] 4f75d¹6s² [Xe] 4f%s2 [Xe] 4f106s2 [Xe] 4f¹¹6s² NRNKERKR [Xe] 4f126s2 [Xe] 4f136s2 [Xe] 4f146s2 [Xe] 4f¹45d¹6s² 72 Hf 73 Ta W 74 75 Re 76 Os 77 Element 78 79 80 81 82 83 84 85 86 87 Fr [Kr] 4d¹05s²5p5 88 Ra [Kr] 4d¹05s²5p6 89 Ac [Xe] 6s¹ 90 Th 91 92 93 94 95 96 97 98 99 Ir Pt Au Hg TI Pb Bi Po At Rn Pa U Np Pu Am Cm Bk Cf Es 100 Fm 101 Md 102 No 103 Lr 104 Rf 105 Db 106 Sg Configuration [Xe] 4f¹45d²6s² [Xe] 4f145d³6s² [Xe] 4f145d46s2 [Xe] 4f145d56s2 [Xe] 4f145d6s2 [Xe] 4f¹45d²6s² [Xe] 4f¹45dº6s¹ [Xe] 4f145d106s1 [Xe] 4f145d106s2 [Xe] 4f145d6s26p* [Xe] 4f145d106s36p? [Xe] 4f145d16s?6p3 [Xe] 4f145d6s®6p* [Xe] 4f145d16s26p5 [Xe] 4f145d106s26p6 [Rn] 7s¹ [Rn] 7s² [Rn] 6d¹7s² [Rn] 6d²7s² [Rn] 5f26d¹7s² [Rn] 5f³6d¹7s² [Rn] 5f46d¹7s2 [Rn] 5f67s² [Rn] 5f77s² [Rn] 5f76d¹7s² [Rn] 5f97s2 [Rn] 5f107,2 [Rn] 5f117s2 [Rn] 5f¹27s² [Rn] 5f137,2 [Rn] 5f147s2 Rn] 5f¹46d¹7s² [Rn] 5f¹46d²7s² [Rn] 5f¹46d³7s2 [Rn] 5f¹46d¹7s²
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)

Answered By

Muhammad Umair
I have done job as Embedded System Engineer for just four months but after it i have decided to open my own lab and to work on projects that i can launch my own product in market. I work on different softwares like Proteus, Mikroc to program Embedded Systems. My basic work is on Embedded Systems. I have skills in Autocad, Proteus, C++, C programming and i love to share these skills to other to enhance my knowledge too.
3.50+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Use data from Appendix C, Figure 7.9, and Figure 7.11 to calculate the lattice energy of RbCl. Is this value greater than or less than the lattice energy of NaCl? Explain. 2372 2081 1312 1681 9 1402...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Use data from Appendix D and other information from this chapter to estimate the temperature at which the dissociation of I 2 (g) becomes appreciable [for example, with the I 2 (g) 50% dissociated...
-
Marcus is the HR manager for United Airlines, an Illinois-based company. One of his employees has recently become disabled and is unable to fulfill the essential functions of his current position,...
-
The following financial statements are available for Sherwood Real Estate Company: Sherwood Company is using these financial statements to entice investors to buy stock in the company. However, a...
-
Let a sample space be partitioned into three mutually exclusive and exhaustive events, B1, B2, and B3. Complete the following probabilitytable. Prior Probabilities PB.) = 0.10 P(B2) Conditional...
-
What the methods are for service of a summons and complaint?
-
Account balances taken from the ledger of High Flying Logistics Co. on December 31, 2011, follow: Cash . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
You have decided to get into the business of selling gourmet brownies. The brownies you will sell can either be made for you, by contracting with a commercial bakery (the buy alternative) OR you can...
-
Despite the fact that it has the higher molecular mass, XeO 4 exists as a gas at 298 K, whereas XeO 3 is a solid. Give a plausible explanation for this observation.
-
One reaction for the production of adipic acid, HOOC(CH 2 ) 4 COOH, used in the manufacture of nylon, involves the oxidation of cyclohexanone, C 6 H 10 O, in a nitric acid solution. Assume that...
-
Refer to the previous exercise. a. Find the P-value for testing whether the population means are equal. Use a two-sided alternative. b. Summarize assumptions for the analysis in part a. Do you think...
-
Nitrogen at \(25 \mathrm{psia}\) and \(850^{\circ} \mathrm{R}\) is flowing at a Mach number of 2.54. After expanding around a smooth convex corner, the velocity of the nitrogen is found to be \(4000...
-
Lenders and investors both provide monetary resources to a company. What do they expect from the company? What are the potential conflicts of interest between lenders and investors?
-
Given that almost each country has its own generally accepted accounting principles, why do we need a set of internationally accepted accounting standards such as IFRS?
-
What are the possible reasons for the SEC's waiver of the U.S. GAAP reconciliation for foreign registrants that followed IFRS to prepare financial statements?
-
What does IFRS stand for? Who sets up the IFRS and are there many countries adopting IFRS? What is the advantage to adopt IFRS?
-
How do the auditors consider the laws and regulations related to an audit client during the audit process?
-
5. How much would you need to deposit in an account now in order to have $5,000 in the account in 5 years? Assume the account earns 2% interest compounded monthly. 10. You deposit $300 each month...
-
Listed here are some items found in the financial statements of Ellyn Toth, Inc. Indicate in which financial statement(s) each item would appear. (a) Service revenue. (b) Equipment. (c) Advertising...
-
A companys net income appears directly on the income statement and the retained earnings statement, and it is included indirectly in the companys balance sheet. Do you agree? Explain.
-
What is the basic accounting equation?
-
Tyler Company has the following information related to purchases and sales of one of its inventory items. Date Description Units Purchased at Cost Units Sold at Retail Sept. 1 Sept. 10 Beginning...
-
How much would an investor be willing to pay for an investment that guaranteed payments of $4,000 at the end of every six months for the next 15 years if they wished to earn a nominal return of...
-
Jackson Company adopted Dollar Value LIFO (DVL) on January 1, 2012 for its one inventory pool. The inventory's value on this date was $500,000. The 2012, 2013 and 2014 ending inventory valued at...

Study smarter with the SolutionInn App


