For the reaction 2 NO 2 (g) 2 NO(g) + O 2 (g), K c =
Question:
For the reaction 2 NO2(g) ⇌ 2 NO(g) + O2(g), Kc = 1.8 x 10-6 at 184 °C. What is the value of Kp for this reaction at 184 °C, for pressures expressed in atmospheres?
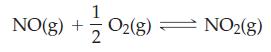
Transcribed Image Text:
(8)702 + NO(g) + O2(g) NO2(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
Python import math Given Kc and the balanced chemical equation calculate Kp ...View the full answer

Answered By

Rodrigo Louie Rey
I started tutoring in college and have been doing it for about eight years now. I enjoy it because I love to help others learn and expand their understanding of the world. I thoroughly enjoy the "ah-ha" moments that my students have. Interests I enjoy hiking, kayaking, and spending time with my family and friends. Ideal Study Location I prefer to tutor in a quiet place so that my students can focus on what they are learning.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
The reaction 2 NO2 2 NO + O2 has the rate constant. k = 0.63 M-1s-1. Based on the units for k, is the reaction first or second order in NO2? If the initial concentration of NO2 is 0.100M, how would...
-
Hugo read a blog about brainstorming and decided that this would be a great way for his customer relations team at Royal Caribbean Cruise lines to generate new ideas for assuring passengers that the...
-
What are two cash management issues for a clothing business? explain Cash Management Issues Working capital management Need for accurate cash flow forecasting Evaluate capital expenditure Optimize...
-
Read each case carefully and, as determined in the Internal Revenue Code of Puerto Rico, identify the deductions, calculate the amount of deductions and what the determined contribution would be. and...
-
Litho-Print is considering two possible capital structures, A and B, shown in the following table. Assume a 40% tax rate. a. Calculate two EBIT-EPS coordinates for each of the structures by selecting...
-
The following information relates to Karen Weigel Co. for the year 2014. Retained earnings, January 1, 2014 .........$48,000 Dividends during 2014 ............. 5,000 Service revenue ...................
-
Assume that Midway Cycles bought and sold a line of mountain bikes during May as follows: Midway Cycles uses the perpetual inventory system. Requirements 1. Compute the cost of ending inventory under...
-
Financial budgets: wages and expense budgets Nathaniel??s Motor Shop does major repair work on automobile engines. The major cost in the shop is the wages of the mechanics. The shop employs nine...
-
Which of the triple constraints do you think is the most important for the project of implementing an enterprise hiring system for MTC ? You should address it from a project - perspective, not the...
-
Continuous removal of one of the products of a chemical reaction has the effect of causing the reaction to go to completion. Explain this fact in terms of Le Chteliers principle.
-
Exactly 1.00 mol each of CO and Cl 2 are introduced into an evacuated 1.75 L flask, and the following equilibrium is established at 668 K. For this equilibrium, calculate (a) The partial pressure of...
-
Which of the following would not appear in the investing section of the statement of cash flows? a. Purchase of inventory. b. Sale of investments. c. Purchase of land. d. All of the above would...
-
How do advanced thermodynamic models, such as the Wilson equation or NRTL (Non-Random Two-Liquid) model, assist in predicting and analyzing the absorption behavior in multi-component systems ?
-
You expected to receive $1,000 today, $1000 one month from now and $1000 two months from now. What is the present value of these cash flows if the dicount rate is 12% APR wit monthly compounding?
-
Ronald Robert has spent much of the last few years gambling and playing soccer in Chicago. He has been largely unemployed during that period. About two years ago, however, he got his act together and...
-
Camila Company has set the following standard cost per unit for direct materials and direct labor. Direct materials (14 pounds @ $4 per pound) Direct labor (2 hours @ $14 per hour) $ 56 28 During...
-
Discuss the role of mass transfer coefficients and the impact of interfacial phenomena in gas-liquid absorption processes. How do hydrodynamic conditions influence the efficiency of mass transfer ?
-
Distinguish between predatory pricing and penetration pricing. Are they the same thing?
-
Design a circuit which negative the content of any register and store it in the same register.
-
Pet Joy Wholesale Inc., a pet wholesale supplier, was organized on May 1, 2010. Projected sales for each of the first three months of operations are as follows: May $360,000 June 450,000 July 600,000...
-
Office Mate Supplies Inc. has cash and carry customers and credit customers. Office Mate estimates that 25% of monthly sales are to cash customers, while the remaining sales are to credit customers....
-
Excel Learning Systems Inc. was organized on May 31, 2010. Projected selling and administrative expenses for each of the first three months of operations are as follows: June $117,400 July 110,500...
-
Joyce Murphy runs a courier service in downtown Seattle. She charges clients $0.43 per mile driven. Joyce has determined that if she drives 3,200 miles in a month, her total operating cost is $1,025....
-
Sandy Bank, Incorporated, makes one model of wooden canoe. And, the information for it follows: Number of canoes produced and sold 550 750 900 Total costs Variable costs $ 104,500 $ 142,500 $ 171,000...
-
The consulting company Allen Hall & Walker (AHW) is in that never-ending budgeting phase of the year. Realizing that they couldn't defer a technology update any longer, the managers plan to replace...

Study smarter with the SolutionInn App


