For the reaction C 2 H 4 (g) + Cl 2 (g) C 2 H 4
Question:
For the reaction C2H4(g) + Cl2(g) → C2H4Cl2(l), determine ΔrH°, given that
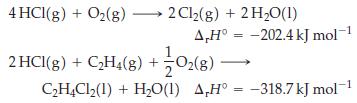
Transcribed Image Text:
4 HCl(g) + O₂(g) →→→→ 2Cl₂(g) + 2 H₂O(1) A,H° -202.4 kJ mol-¹ 2HCl(8) + C₂H4(8) + O2(8) - = C₂H4Cl₂(1)+ H₂O(1) AH° -318.7 kJ mol-1 =
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
To determine rH for the reaction C2H4g Cl2g C2H4Cl2l we can use the ...View the full answer

Answered By

Ayush Jain
Subjects in which i am expert:
Computer Science :All subjects (Eg. Networking,Database ,Operating System,Information Security,)
Programming : C. C++, Python, Java, Machine Learning,Php
Android App Development, Xamarin, VS app development
Essay Writing
Research Paper
History, Management Subjects
Mathematics :Till Graduate Level
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Which is a combustion reaction and which is not? a. C2H4 + O2 ( C2H4O2 b. C2H4 + Cl2 ( C2H4Cl2
-
The solubility of Cl2 in 100 g of water at STP is 310 cm3. Assume that this quantity of Cl2 is dissolved and equilibrated as follows: (a) If the equilibrium constant for this reaction is 4.7 Ã...
-
Determine H for the reaction 2CH4 ( 2H2 + C2H4 Given these data: CH4 + 2O2 ( CO2 + 2H2O H = 891 kJ C2H4 + 3O2 ( 2CO2 + 2H2O H = 1,411 kJ 2H2 + O2 ( 2H2O H = 571 kJ
-
Attijari Wafa Bank distributed on May 2022 a dividend per share of 5 MAD / share. The company is planning to have next 3 years a growth rate of 5% then after 7%. If the required rate of return is 12%...
-
In the following chart, compare financial accounting and managerial accounting by describing how each differs for the items listed. Be specific in yourresponses. Flnancial Accounting Managerial...
-
Company XYZs capital adequacy over the last three years, as measured by the three key capital ratios, signals conditions that are: A . mixed. B . declining. C . improving.
-
SWIGART v. BRUNO CALIFORNIA COURT OF APPEALS 13 CAL. APP. 5TH 529 2017 According to the American Endurance Ride Conference, endurance riding is a highly competitive and demanding sport. It is...
-
Bertha's Bathworks produces hair and bath products. Its biggest customer is a national retail chain that specializes in such products. Bertha Jackson, the owner of Bertha's Bathworks, would like to...
-
13. An analyst is valuing Red Inc. common stock using the dividend discount model. The company plans to start paying dividends with its first dividend of $3.25 per share occurring next year. To...
-
Prepare the journal entries of the following transactions. 1. On January 1, 20XX, BVD Corp. issued 3,500 shares of $50 par, 7% preferred stock for $275,000. 2. On January 1, 20XX, BVD Corp. also...
-
Do you think the following observation is in any way possible? An ideal gas is expanded isothermally and is observed to do twice as much work as the heat absorbed from its surroundings. Explain your...
-
In an adiabatic process, a system is thermally insulatedthere is no exchange of heat between system and surroundings. For the adiabatic expansion of an ideal gas (a) Does the gas do work? (b) Does...
-
Change the numbers from scientific notation to ordinary notation. 8 10 0
-
You are required to make a choice among three alternatives for a distribution system for your firm. The first option involves investment of $30 billion to build a new distribution system costing $30...
-
1b) Who are the duty holders and their responsibilities (according to WHS Laws) in regard to implementing the organisation's WHSMS? 1c) Section 19(3)c of the WHS Act 2011 states a PCBU must provide...
-
A new network was set up with the Internet protocol (IP) range of 192.168.13.2-192.168.13.100. The internet service provider has set up a static IP address of 104.215.148.63. How are devices able to...
-
Narrative Techniques - Atwood uses a variety of narrative techniques in the telling of her story. Find an example of each of the following techniques in the novel. Technique Example 1st person...
-
A traffic engineer counted vehicles on Rt. 9 and found that, on average, a vehicle passes in front of her at a rate of every 5 seconds. If vehicles keep coming at this rate, what would be the...
-
Why is misstatement of inventory one of the most common means of financial statement fraud?
-
For all of the following words, if you move the first letter to the end of the word, and then spell the result backwards, you will get the original word: banana dresser grammar potato revive uneven...
-
Accounting Change and Error Analysis on December 31, 2010, before the books were closed, the management and accountants of Madras a Inc. made the following determinations about three depreciable...
-
Error Corrections you have been assigned to examine the financial statements of Zarle Company for the year ended December 31, 2010. You discover the following situations. 1. Depreciation of $3,200...
-
Comprehensive Error Analysis on March 5, 2011, you were hired by Hemingway Inc., a closely held company, as a staff member of its newly created internal auditing department. While reviewing the...
-
Suppose that the estimated value of a loyal customer is $80 per year. The gross margin is 50%. Customers but the product every 2 years. Assume that the defection rate, r, is given by 17%. Compute the...
-
What are the trade-offs and constraints that govern the evolution of adaptive traits, including physiological, morphological, and behavioral adaptations, and how do these trade-offs impact the...
-
How do coevolutionary interactions, such as host-parasite interactions, predator-prey dynamics, and mutualistic relationships, drive reciprocal adaptation and promote the diversification of...

Study smarter with the SolutionInn App


