Give the major products obtained in each of the following reactions and indicate which mechanisms are involved:
Question:
Give the major products obtained in each of the following reactions and indicate which mechanisms are involved:
(a)
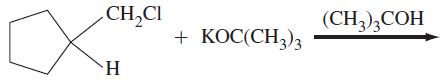
(b)
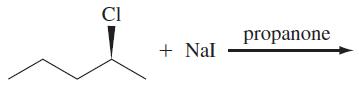
(c)
![]()
Transcribed Image Text:
CH₂Cl H + KOC(CH3)3 (CH3)3COH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Determine A in the indicated figures. Fig. 2.40 (c) B 4 66 A (c) 4 C
-
Give the major products obtained in each of the following reactions and indicate which mechanisms are involved: (a) (b) (c) Br + NaNH, NH3(1)
-
Give the major products of each of the following reactions a. b. c. d. CHa NCH3 CH3 HO CH3CH2CH2NCH CH H3C CH3 H3C - H3C CH3
-
Express the given quantity in terms of sin x and cos x. sin 2 X
-
The following data are available for Haul-It-Away Truckers: 1. Compute the predetermined overhead rate for each of the two years, if based on (a) Direct labor hours, (b) Number of moving jobs, and...
-
Why is there an incentive for a manager to inappropriately reduce reported profit when it appears that profits are likely to be above the upper limit of a managers bonus range?
-
1. Read the text materials on feedforward, concurrent, and feedback control. 2. Read the Control Problem Situation and be prepared to resolve those control problems in a group setting. 3. Your...
-
Three different plans for financing an $80,000,000 corporation are under consideration by its organizers. Under each of the following plans, the securities will be issued at their par or face amount,...
-
Two large conducting plates on insulating stands are placed a distance D = 50 cm apart, as shown at right. The inner surface of one has a charge density of +0; the other, - The charge density on the...
-
A sample of (R)-CH 3 CH 2 CH(CH 3 )Cl reacts with CH 3 O in dimethyl sulfoxide, (CH 3 ) 2 SO, a convenient solvent for organic reactions. The resulting solution is optically active. (a) Write the...
-
A sample of (S)-CH 3 CH 2 CH(CH 3 )Cl is hydrolyzed by water, and the resulting solution is optically inactive. (a) Write the formula of the product. (b) By which nucleophilic substitution reaction...
-
The following table indicates the number of constitutional isomers with molecular formula C 7 H 16 . Draw each of the isomers, making sure not to draw the same compound twice. NUMBER OF...
-
Name the following compound: PBr3 Phosphorous bromide Potassium bromide Phosphorous (III) bromide Phosphorous tribromide Potassium(III) bromide Potassium tribromide 0 0 0 0 0
-
When 0.560 g of NA reacts with excess f2 to form NAf 12.8 kk of heat is evolved at standard state conditions
-
In a laboratory setting, concentrations for solutions are measured in molarity, which is the number of moles per liter ( mol / L ) . Concentrations are often converted to more common units on the...
-
nitrogen - 1 3 undergoing positron emission Express your answer as a nuclear equation.
-
A certain liquid X has a normal freezing point of 4 . 2 0 \ deg C and a freezing point depression constant = Kf 4 . 9 7 \ deg C kgmol 1 . A solution is prepared by dissolving some ammonium sulfate (...
-
Explain how worker's compensation laws benefit both employers and employees but in different ways.
-
10m solution. If Ka(HA) = 10 then pOH of solution will be [Given : log4=0.6] (A) 6.7 (B) Greater than 6.7 & less than 7.0 (C) Greater 7.0 & less than 7.3 (D) Greater than 7.3
-
Define the term capital intensity. Explain how a decline in capital intensity would affect the AFN, other things held constant. Would economies of scale combined with rapid growth affect capital...
-
Define the term self-supporting growth rate. Based on the Figure MC-1 data, what is Hatfields self-supporting growth rate? Would the self-supporting growth rate be changed by a change in the capital...
-
How much new capital will the firm need (i.e., what is the forecasted AFN), how does it compare with the amount you calculated using the AFN equation, and why does any difference exist?
-
GeneralRobotics,Inc.,alarge and well-known company, is about to issue$5,000,000worth ofcommercialpaper. The paper hasamaturity ofsix months(182days),and the market iswillingto pay97%of parvaluefor...
-
The last person to use the rotovap took off acetone. You need to take off ethanol. Should you check the receiving flask, and why should you? If a small amount of acetone is left in the receiving...
-
If Airsonics purchases the helicopter, before tax operating cost will be $200,000 per year, payable at the beginning of each year. After 6 years helicopter will be worth either $400,000 or $600,000....

Study smarter with the SolutionInn App


