Given the equilibrium constant values 1 N2(g) + O2(g) = NO(g) K = 2.7 10-18 2
Question:
Given the equilibrium constant values
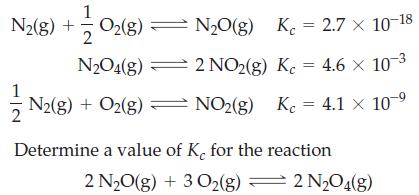
Transcribed Image Text:
1 N2(g) + O2(g) = N₂O(g) K₁ = 2.7 × 10-18 2 NO2(g) Kc = 4.6 x 10-³ Kc = 4.1 × 10-⁹ N2O4(g) N2(g) + O2(g) — NO₂(g) N2(g) + O2(g) 2 Determine a value of K, for the reaction 2 N₂O(g) + 3 O₂(g) = 2 N₂O4(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 25% (4 reviews)
Given the equilibrium constant values Kc1 27 1018 for the reaction N2g O2g ...View the full answer

Answered By

Jehal Shah
I believe everyone should try to be strong at logic and have good reading habit. Because If you possess these two skills, no matter what difficult situation is, you will definitely find a perfect solution out of it. While logical ability gives you to understand complex problems and concepts quite easily, reading habit gives you an open mind and holistic approach to see much bigger picture.
So guys, I always try to explain any concept keeping these two points in my mind. So that you will never forget any more importantly get bored.
Last but not the least, I am finance enthusiast. Big fan of Warren buffet for long term focus investing approach. On the same side derivatives is the segment I possess expertise.
If you have any finacne related doubt, do reach me out.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Wallace and Brace CPA assign a partner to screen and recommend (or decline) potential clients prior to acceptance. Discuss at least 4 specific issues which should have been considered prior to...
-
A liquid mixture containing 70.0 mol of n -pentane and 30.0 mol of n -hexane initially at 46?C is partially vaporized at P = 1 atm in a single-stage distillation apparatus (Rayleigh still). The heat...
-
Wayne University sent a self-audit questionnaire to their employees to get an overview of how their staff viewed the internal control procedures. Internal audits are responsible for examining risks...
-
Taft Corporation operates primarily in the United States. However, a few years ago it opened a plant in Spain to produce merchandise to sell there. This foreign operation has been so successful that...
-
The Environmental Protection Agency sometimes imposes penalties on firms that pollute the environment. But did you know that there is a legal market for pollution? A mechanism that has been developed...
-
A stepped shaft ACB having solid circular cross sections with two different diameters is held against rotation at the ends (see figure). If the allowable shear stress in the shaft is 6000 psi, what...
-
MPS Industries is considering the purchase of equipment costing $100,000.00 having a seven-year useful life. Projected net cash flows from the investment are shown below. Determine the net present...
-
You sell bicycle theft insurance. If bicycle owners do not know whether they are high- or low risk consumers, is there an adverse selection problem?
-
Think about a class you have taken online in the past or present. In what ways can instructors leverage Emotional Response Theory in online class settings? What strategies can the instructors...
-
Solid silver is added to a solution with these initial concentrations: [Ag + ] = 0.200 M, [Fe 2+ ] = 0.100 M, and [Fe 3+ ] = 0.300 M. The following reversible reaction occurs. What are the ion...
-
(A) If 0.150 mol H 2 (g) and 0.200 mol I 2 (g) are introduced into a 15.0 L flask at 445 C and allowed to come to equilibrium, how many moles of HI(g) will be present? (B) Suppose the equilibrium...
-
Farnsworth Drycleaners has capacity to clean up to 7,500 garments per month. Requirements 1. Complete the following schedule for the three volumes shown. 2. Why does the average cost per garment...
-
What steps should an ethical leader take to protect the organization from risk? Which kind of threat is most detrimental to a nonprofits good standing in the community? Why do you believe this to be...
-
Schein and Van Maanen (2016) uses the term career anchors to refer to individual preferences regarding work. Do you believe dedication to a cause is still the primary reason people choose to work in...
-
If you believe faith- based nonprofits merit special consideration in competing for government support, what expectations of these organizations are justified in return? If you feel special...
-
Is the role of ethical or moral compass of the organization harder today than in the past for a nonprofit leader? Justify your position.
-
Based on the information provided in the featured case study, Youth Service Networks founder, Trevor Clinton, likely cannot be considered a servant- leader. Which of his actions seem most at odds...
-
Assume that 10% of us believe that college is not a good investment (based on a survey in USA Today). a. Let A denote the event of selecting someone who believes that college is not a good...
-
For each of the following transactions, indicate whether it increases, decreases, or has no effect on the following financial ratios: current ratio, debt-to-equity ratio, profit margin ratio, and...
-
Hudson Company operates two factories. The company applies factory overhead to jobs on the basis of machine hours in Factory 1 and on the basis of direct labor hours in Factory 2. Estimated factory...
-
Willies Engine Shop uses a job order cost system to determine the cost of performing engine repair work. Estimated costs and expenses for the coming period are as follows: Engine parts $ 875,000 Shop...
-
The Medical Center has a single operating room that is used by local physicians to perform surgical procedures. The cost of using the operating room is accumulated by each patient procedure and...
-
TABLE 1 Present Value of $1 n 4% 0.961538 11234567890- 10 6% 0.943396 16% 20% 5% 7% 8% 9% 10% 12% 14% 0.952381 0.934579 0.925926 0.917431 0.909091 0.892857 0.877193 0.862069 0.833333 0.924556...
-
Hanmi Group, a consumer electronics conglomerate, is reviewing its annual budget in wireless technology. It is considering investments in three different technologies to develop wireless...
-
Find standard deviation and average: (show me the steps.) 4.5 x 10^8 cells/mL 2.55 x 10^6 cells/mL 8.50 x 10^5 cells/mL 2.3 x 10^5 cells/mL 3.605 x 10^6 cells/mL 4.71 x 10^5 cells/mL 3.63 x 10^6...

Study smarter with the SolutionInn App


