How much heat, in kilojoules, is associated with the production of 283 kg of slaked lime, Ca(OH)
Question:
How much heat, in kilojoules, is associated with the production of 283 kg of slaked lime, Ca(OH)2?
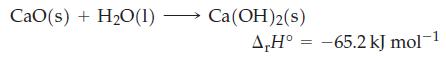
Transcribed Image Text:
CaO(s) + H₂O(1) - Ca(OH)2(s) A,H° -65.2 kJ mol-1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
To find the heat associated with the production of 283 kg of slaked lime CaOH2 from calcium oxide Ca...View the full answer

Answered By

Oleksandr Kinash
I have a Master's degree in International Economics. I have been working as a tutor and academic research writer for over 5 years now. I mostly help students is Economics, Statistics, and Math.
0.00
0 Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
A company is considering a 5-year project that opens a new product line and requires an initial outlay of $77,000. The assumed selling price is $98 per unit, and the variable cost is $60 per unit....
-
Find ATA and AAT. A = [20 3 57 11-3 3 82 0-4 40 0-2 (a) ATA (b) AAT 11
-
What is the classification of the freight in account?
-
A data analyst decided to do all design work from home and wants to create a home office. The analyst needs a new computer for $1,900 and printer/scanner for $250. A vendor offers a financing option...
-
List the four components of a manufacturing statement and provide specific examples of each for Apple.
-
A skydiver has his parachute open and is floating downward through the air at a constant speed. Describe a situation. For each problem, identify all the forces acting on the object and draw a...
-
Ormet Primary Aluminum Corporation, operated an aluminum smelter plant in Hannibal, Ohio. The facility ceased production in October 2013 in order to liquidate its assets after filing for bankruptcy...
-
Express Distribution markets CDs of the performing artist Fishe. At the beginning of October express had in beginning inventory 2,000 of Fishes CDs with a unit cost of $7. During October, Express...
-
A bug sits on a record moving at a constant angular speed of 0 = 3.5 rad/s. Starting at t = 0 s, the record player is turned off and is given a constant angular acceleration of = 1.0 rad/s 2 . The...
-
On January 1, 2024, Harlon Consulting entered into a three-year lease for new office space agreeing to lease payments of $5,000 in 2024, $6,000 in 2025, and $7,000 in 2026. Payments are due on...
-
What is the final temperature (in C) of 1.24 g of water with an initial temperature of 20.0 C after 6.052 J of heat is added to it?
-
Brass has a density of 8.40 g/cm 3 and a specific heat capacity of 0.385 J g-1 C -1 . A 15.2 cm 3 piece of brass at an initial temperature of 163 C is dropped into an insulated container with 150.0 g...
-
Which API provides access to properties and methods related to the device battery? a. Geolocation API b. Battery Status API c. Device Orientation API d. WebRTC API
-
Compare and contrast predictive (waterfall) value delivery with Agile delivery. How does the organization benefit from each of these value delivery methods? Is it possible to deliver value in...
-
Check that 1 tan(x/2) = 1 + tan a 1 and sin(x/2)= sin x 1+ tan (1/2) holds for any x (-/2,/2)\{0}. Use these relations to write a program in Python computing the fifth first decimal of T with the...
-
Compare between American management vs Japanese management. seven paragraphs in own wording, Compare/contrast the management styles of American management Compare/contrast employee work attitudes...
-
How do sociological theories such as structural functionalism, conflict theory, and symbolic interactionism help us understand and analyze various social phenomena such as deviance, social...
-
2. During January 2024, the records of the company were inadequate. You were asked to prepare the summary of the preceding transactions. To develop a quick assessment of their economic effects on...
-
Record each of the following transactions using T accounts, assuming the perpetual inventory system is used: Aug. 2 Purchased merchandise on credit from Vera Company, invoice dated August 1, terms...
-
The following selected accounts and normal balances existed at year-end. Notice that expenses exceed revenue in this period. Make the four journal entries required to close the books: Accounts...
-
Cominsky Company purchased a machine on July 1, 2011, for $28,000. Cominsky paid $200 in title fees and county property tax of $125 on the machine. In addition, Cominsky paid $500 shipping charges...
-
Dickinson Inc. owns the following assets. Compute the composite depreciation rate and the composite life of Dickinson's assets. Estimated Useful Life 10 years Cost Asset Salvage A $70,000 $ 7,000...
-
Holt Company purchased a computer for $8,000 on January 1, 2009. Straight-line depreciation is used, based on a 5-year life and a $1,000 salvage value . In 2011, the estimates are revised. Holt now...
-
Suppose that a firm manufactures wallets. The firm's fixed cost is $106560, and the variable cost is $3 per wallet. If each wallet sells for $65, how many wallets need to be sold in order for the...
-
You are attempting to value a call option with an exercise price of $80 and one year to expiration. The underlying stock pays no dividends, its current price is $80, and you believe it has a 50%...
-
Stocks X and Y have the following probability distributions of expected future returns: Probability 0.1 X Y (10%) (35%) 0.2 0.4 0.2 2224 0 12 20 20 25 0.1 48 55 What are the expected returns of both...

Study smarter with the SolutionInn App


