How much heat is required to raise the temperature of 215 g CH 3 OH(l) from 20.0
Question:
How much heat is required to raise the temperature of 215 g CH3OH(l) from 20.0 to 30.0 °C and then vaporize it at 30.0 °C? Use data from Table 12.4 and a molar heat capacity of CH3OH(l) of 81.1 J mol-1K-1.
Table 12.4
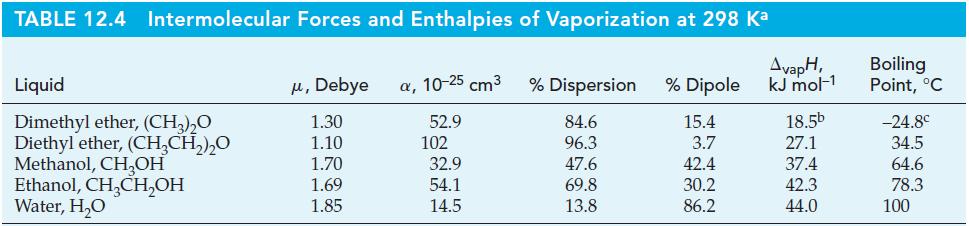
Transcribed Image Text:
TABLE 12.4 Intermolecular Forces and Enthalpies of Vaporization at 298 Ka Liquid Dimethyl ether, (CH₂)₂O Diethyl ether, (CH₂CH₂)₂0 Methanol, CH₂OH Ethanol, CH₂CH₂OH Water, H₂O μ, Debye α, 10-25 cm³ 1.30 52.9 1.10 1.70 1.69 1.85 102 32.9 54.1 14.5 % Dispersion % Dipole 84.6 15.4 96.3 3.7 47.6 42.4 69.8 30.2 13.8 86.2 AvapH, kJ mol-1 18.5b 27.1 37.4 42.3 44.0 Boiling Point, °C -24.8⁰ 34.5 64.6 78.3 100
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (3 reviews)
Step 1 Heating the liquid from 200 to 300 C Heat required q1 mass m specific heat capacity c te...View the full answer

Answered By

Albert Kinara
i am an expert research writer having worked with various online platform for a long time. i also work as a lecturer in business in several universities and college part time and assure you well researched and articulate papers. i have written excellent academic papers for over 5 year and have an almost similar experience experting many clients in different units. bachelor of commerce (finance)
masters in strategic management
phd finance
4.60+
26+ Reviews
48+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Debye's T3 Law. At very low temperatures the molar heat capacity of rock salt varies with temperature according to Debye's T3law: C= kT3/O3 Where k = 1940J/mol' K and 0= 281 K. (a) How much heat is...
-
5 Match the items in List-I with items in List-II. List-I List-II a) Margin of Safety i) Earning Power b) ROI ii) Cash Flow statement c) Current Ratio iii) Break Even analysis d) Cash Equivalentsiv)...
-
A 0.400-kg aluminum teakettle contains 2.00 kg of water at 15.0C. How much heat is required to raise the temperature of the water (and kettle) to 100.0C?
-
The rigid bar AB is supported by a pin at B and by two the cables AC (perpendicular to the beam) and AD (inclined with respect to the beam) attached at A as shown in Fig. 3. A C 30 in D B 80 in...
-
United Road Machinery Company, a dealer in heavy road equipment (including truck scales supplied by Thurman Scale Company), received a telephone call on July 21 from James Durham, an officer of...
-
What is ITSM?
-
How can new methods of promotion through the internet assist the sales process?
-
(Rule-Making Issues) When the FASB issues new pronouncements, the implementation date is usually 12 months from date of issuance, with early implementation encouraged. Karen Weller, controller,...
-
Program development involves learning and understanding the major principles and models of budgeting. Budgeting for financial control of a human services program involves making decisions about...
-
How many liters of CH 4 (g), measured at 23.4 C and 768 mmHg, must be burned to provide the heat needed to vaporize 3.78 L of water at 100 C? For CH 4 , comb H = -8.90 x 10 2 kJ mol -1 . For H 2...
-
A vapor volume of 1.17 L forms when a sample of liquid acetonitrile, CH 3 CN, absorbs 1.00 kJ of heat at its normal boiling point (81.6 C and 1 atm). What is vap H in kilojoules per mole of CH 3 CN?
-
Explain the difference between committed and discretionary fixed costs. Give examples of each.
-
This assignment is intended to help you build a supply chain plan/diagram for a new business by analyzing factors that affect sourcing, logistics, metrics, suppliers, and risk. Create a diagram of...
-
Provide recommendations for the Walt Disney company and how they can continue to expand?
-
On January 1, 2XX1, Bargain Inc. acquired 100% of Wind Corporation for $9,750,000 cash. On that date, Willey's total stockholders' equity was $7,250,000. The following assets had fair values...
-
Explain what are the benefits that companies are federally encouraged to provide?
-
What are the differences between the churches of the 18th century and 19th to the churches today?
-
What events resulted in banks shift from the traditional banking model of originate and hold to a model of originate and distribute?
-
Write a program to move a signed number from smaller register to bigger register. Hint: movzx ax, bl Topic: Data Related Operators and Directives in assembly language
-
Payback Period concerning payback: a. Describe how the payback period is calculated, and describe the information this measure provides about a sequence of cash flows. What is the payback criterion...
-
Discounted Payback Concerning discounted payback: a. Describe how the discounted payback period is calculated, and describe the information this measure provides about a sequence of cash flows. What...
-
Average Accounting Return Concerning AAR: a. Describe how the average accounting return is usually calculated, and describe the information this measure provides about a sequence of cash flows. What...
-
Taveras Corporation is currently operating at 50% of its available manufacturing capacity. It uses a job-order costing system with a plantwide predetermined overhead rate based on machine-hours. At...
-
Qinto Company sells two types of products: basic and deluxe. The company provides technical support for its products at a budgeted overhead cost of $239,200 per year. The company allocates technical...
-
Anderson Corporation has found that 8 0 % of its sales in any given month are credit sales, while the remainder are cash sales. Of the credit sales, Anderson Corporation has experienced the following...

Study smarter with the SolutionInn App


