Identify any chiral carbon atoms in the molecules to the right. CH3 Methylcyclohexane CH34 CH3 cis-1,3-dimethylcyclohexane
Question:
Identify any chiral carbon atoms in the molecules to the right.
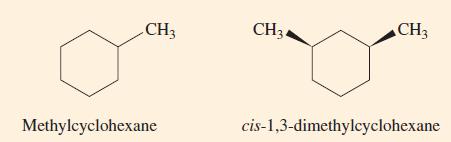
Transcribed Image Text:
CH3 Methylcyclohexane CH34 CH3 cis-1,3-dimethylcyclohexane
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Analyze A carbon atom is chiral if it is bonded to four different groups To determine whether a carb...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
(A) Identify the chiral carbon atoms in the molecule shown in the diagram on the right. (B) How many chiral atoms are there in 1,1,3-trimethylcyclohexane?
-
When Alfred Werner was developing the field of coordination chemistry, it was argued by some that the optical activity he observed in the chiral complexes he had prepared was because of the presence...
-
In 1994 chemists at Texas A&M University reported the synthesis of a non-naturally occurring amino acid: a. To which naturally occurring amino acid is this compound most similar? b. A tetrapeptide,...
-
A cancerous tumor is modeled as a sphere of radius r cm. a. At what rate is the volume V = 4/3r3 changing with respect to r when r = 0.75 cm? b. Estimate the percentage error that can be allowed in...
-
Using the following information, compute the amount of cash from financing activities: 1. Anderson Company purchased $12,000 of its own common stock to be held in the treasury. 2. Anderson paid cash...
-
(a) In Table, note that there are two index funds based on the S&P 500 Index. Suggest a reason why Joshua should invest in one or the other, noting that the returns for the Vanguard 500 Index Fund...
-
Reconsider the data from Problem 4. Management has expressed some concern over the life of the project and the impact of possible early termination. As a result, you have developed additional data...
-
The University Bookstore at a prestigious private university buys mechanical pencils from a wholesaler. The wholesaler offers discounts for large orders according to the following price schedule:...
-
Explain how specific institutions of capitalism can harness self interest to benefit the common good and improve economic outcomes in society at large. Explain what a social dilemma is and how it...
-
If you could measure the individual heats of combustion of axial and equatorial methylcyclohexane, which conformer would burn more exothermically?
-
(A) Which of the following chlorofluorohydrocarbons is chiral: (a) CF 3 CH 2 CCl 3 ; (b) CF 2 HCHFCCl 3 ; (c) CClFHCHHCCl 2 F? (B) Which of the following chloroalcohols is chiral: (a) CH 2 ClCH 2 CH...
-
Why is a data warehouse created as a separate data store?
-
A mass spectrometer is a mass spectrometer device like the one shown in the attached figure. A particle of total charge total q = 4.8-10-19 C is directed with velocity of magnitude v = 4.5 - 106 m/s...
-
The stresses shown in the figure below act at a point on the free surface of a stressed body. Determine the normal stresses and the shear stress if the element oriented -36 from the position shown....
-
Jamal has been the owner of a car dealership for 10 years. One day, Jamal sold one of his most expensive cars to Abdul. At the time of the sale, Jamal thought Abdul acted in a peculiar manner, but he...
-
1. Define temperature. Name the four scales used to measure temperature. 2. Fill in the table below with the appropriate temperatures for water. Boiling Freezing Absolute Zero Kelvin Celsius Rankine...
-
You bought a revenue property on March 1, 2020. You paid $386,000 for the property and put 20% down. You took out a mortgage with a 25 year amortization and an initial 5-year fixed interest rate of...
-
Dr. Gregory B. Nazar removed a malignant tumor from Roe Branhams brain on February 27, 2000. The surgery was a success, but shortly afterward Branham began to experience pain in his head. The pain...
-
Time Travel Publishing was recently organized. The company issued common stock to an attorney who provided legal services worth $25,000 to help organize the corporation. Time Travel also issued...
-
Harley Motors has $10 million in assets, which were financed with $2 million of debt and $8 million in equity. Harleys beta is currently 1.2, and its tax rate is 40%. Use the Hamada equation to find...
-
Firms HL and LL are identical except for their leverage ratios and the interest rates they pay on debt. Each has $20 million in assets, has $4 million of EBIT, and is in the 40% federal-plus-state...
-
The Weaver Watch Company sells watches for $25, the fixed costs are $140,000, and variable costs are $15 per watch. a. What is the firms gain or loss at sales of 8,000 watches? at 18,000 watches? b....
-
How would you weigh the benefits of living in a green, innovative, and hyperconnected city against the costs of being increasingly surveilled and having your data collected and used by governments...
-
3. Wheat is produced in Kansas and lowa and is transported to California on west coast and New York state on east coast. Kansas produces 15000 tons of wheat and lowa Produces 10000 tons of wheat....
-
Consider the following context-free grammars (CFG). With the help of leftmost derivation decide whether the grammars are ambiguous or not. a) E S 0E44 0A E+E | A | F | A 5A7 | 2S3 | 0S4 | & F 5 (5)...

Study smarter with the SolutionInn App


