In the drawing it shown that, 1.00 g H 2 (g) at 300 K is maintained at
Question:
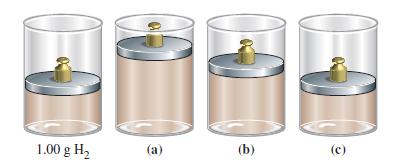
In the drawing it shown that, 1.00 g H2(g) at 300 K is maintained at 1 atm pressure in a cylinder closed off by a freely moving piston. Which sketch, (a), (b), or (c), best represents the mixture obtained when 0.50 g H2(g) is added and the temperature is reduced to 275 K? Explain your answer.
Transcribed Image Text:
1.00 g H₂ (a) (b) (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
The correct answer is b When 050 g H2g is added to the cylinder the total ...View the full answer

Answered By

Shehar bano
I have collective experience of more than 7 years in education. my area of specialization includes economics, business, marketing and accounting. During my study period I remained engaged with a business school as a visiting faculty member and did a lot of business research. I am also tutoring and mentoring number of international students and professionals online for the last 7 years.
5.00+
4+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Can you think of a soft drink besides Pepsi that has the potential to outsell Coca Cola Explain your answer How can Pepsi compete with Coke Is there a market niche the company has not targeted that...
-
In the drawing below, 1.00 g H 2 (g) is maintained at 1 atm pressure in a cylinder closed off by a freely moving piston. Which sketch, (a), (b), or (c), best represents the mixture obtained when 1.00...
-
A gas mixture consists of 320 mg of methane, 175 mg of argon, and 225 mg of neon. The partial pressure of neon at 300 K is 8.87 kPa. Calculate (a) The volume and (b) The total pressure of the mixture.
-
Use the percentages in Figure 7.3 and the values in Figure 7.4 to answer the following questions: What percentage of SAT takers score between 500 and 600? Figure 7.3 Normal Curve Percentages, to Two...
-
Three students share a house. Having better things to do than clean house, they hire someone to come in and clean once each week. How should they share the costs of the housekeeper? One simple...
-
To stimulate the sales of its Alladin breakfast cereal, Loptien Company places 1 coupon in each box. Five coupons are redeemable for a premium consisting of a childs hand puppet. In 2015, the company...
-
FRAUD PREVENTION AND DETERRENCE IN ACTION Assume the following facts: thirty-day collection period. The invoice is used to post-sales to the accounting system. Checks are received in the mailroom and...
-
A project to develop technology training seminars is 5 days behind schedule at day 65. It had a planned cost of $735,000 for this point in time, but the actual cost is only $550,000. Estimate the...
-
The KLM Partnership, which uses the accrual method of accounting, is owned equally by Karen ( cash method taxpayer ) , and LM corporation ( accrual method taxpayer ) . Karen is a real estate...
-
A 2.35 L container of H 2 (g) at 762 mmHg and 24 C is connected to a 3.17 L container of He(g) at 728 mmHg and 24 C. After mixing, what is the total gas pressure, in millimeters of mercury, with the...
-
A gas cylinder of 53.7 L volume contains N 2 (g) at a pressure of 28.2 atm and 26 C. How many grams of Ne(g) must we add to this same cylinder to raise the total pressure to 75.0 atm?
-
Answer the following questions about specific speed and pump similarity. (a) Using both U.S. and Sl units, show that the shape number given in Equation 5.24 is dimensionless. (b) Is specific speed a...
-
An equipment acquisition proposal is being considered by a large healthcare organization, XYZ Health Care. The array machine will enable the hospital to perform autoimmunity tests (for...
-
Amazon is the world's leading online retailer and it's success has spurred other physical,brick,and mortar retailers to have an online presence. It is often referred to as the online equivalent of...
-
Here's the details of Lhean Lim's Citibank credit card statement: Current statement date October 8, 2020 Next statement date November 8, 2020 Previous balance Php 80,000.00 Payment due date October...
-
The dead-weight loss from an excise tax a. is greater if demand is perfectly inelastic. b. is caused by a shift in consumer preferences when the tax is raised. c. is the lost surplus that results...
-
As the nations leading consumer electronics retailer, Best Buy is trying to be the best. But thats not been easy in light of the challenges its facing in the external environment. Like many other...
-
You are the manager of a firm that competes against four other firms by bid-ding for government contracts. While you believe your product is better than the competition, the government purchasing...
-
Government is advised to tax goods whose demand curves are inelastic if the goal is to raise tax revenues. If the goal is to discourage consumption, then it ought to tax goods whose demand curves are...
-
Archer Inc. issued $4,000,000 par value, 7% convertible bonds at 99 for cash. If the bonds had not included the conversion feature, they would have sold for 95. Prepare the journal entry to record...
-
Petrenko Corporation has outstanding 2,000 $1,000 bonds, each convertible into 50 shares of $10 par value common stock. The bonds are converted on December 31, 2010, when the unamortized discount is...
-
Pechstein Corporation issued 2,000 shares of $10 par value common stock upon conversion of 1,000 shares of $50 par value preferred stock. The preferred stock was originally issued at $60 per share....
-
The irreversible zero-order reaction A B is taking place inside the porous catalyst slab shown in Fig 1. [A]o [A]0 [A] = f(x) 2L X FIGURE 1 (a) Show that the concentration profile of reactant A in...
-
Gas-phase catalytic hydrogenation is used to convert o-cresol to 2-methylcyclohexanone (used in fragrances) over a Ni-on-silica catalyst at 170 C in an isothermal packed bed reactor: OH & CH3 CH3 2H2...
-
A steel column circular in cross-section is 150mm in diameter and carries a load of 1.4MN.Calculate a. The compressive stress in the column. b. The plan dimensions of the concrete foundation so that...

Study smarter with the SolutionInn App


