In the following diagrams, which representation demonstrates a strong electrolyte? Gas Aqueous (a) Gas Aqueous Gas Aqueous
Question:
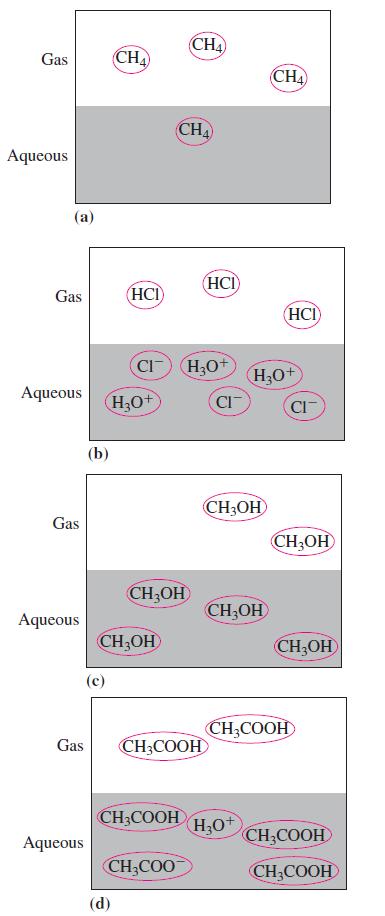
In the following diagrams, which representation demonstrates a strong electrolyte?
Transcribed Image Text:
Gas Aqueous (a) Gas Aqueous Gas Aqueous (b) Aqueous (c) CHA (HCI) H₂O+ CH₂OH (d) CI) H₂0+ CH₂OH CHA Gas CH3COOH CHA CH3COOH CH₂COO HCI CI CH₂OH CH₂OH CHA H₂O+ H₂O+ (HCI) CIT CH₂OH CH₂OH CH₂COOH CH3COOH CH₂COOH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
Among the given representations representation b demonstrates a strong electrolyte In t...View the full answer

Answered By

PALASH JHANWAR
I am a Chartered Accountant with AIR 45 in CA - IPCC. I am a Merit Holder ( B.Com ). The following is my educational details.
PLEASE ACCESS MY RESUME FROM THE FOLLOWING LINK: https://drive.google.com/file/d/1hYR1uch-ff6MRC_cDB07K6VqY9kQ3SFL/view?usp=sharing
3.80+
3+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
In the following diagrams, which representation demonstrates a weak electrolyte? Gas Aqueous Gas Aqueous Gas Aqueous Gas Aqueous (a) (b) CH4 (HCI (d) HO CH3OH CHOH CI-HO+ CHCOOH CH3COOH CHCOO CHA CHA...
-
Case Study: Quick Fix Dental Practice Technology requirements Application must be built using Visual Studio 2019 or Visual Studio 2017, professional or enterprise. The community edition is not...
-
Give three example solutions that fit each of the following descriptions. a. A strong electrolyte solution that is very acidic b. A strong electrolyte solution that is slightly acidic c. A strong...
-
True or False The slope of the line 2y = 3x + 5 is 3.
-
Wallowa Company is considering a long-term investment project called ZIP. ZIP will require an investment of $120,000. It will have a useful life of 4 years and no salvage value . Annual revenues...
-
You have observed the following returns over time: Assume that the risk-free rate is 6% and the market risk premium is 5%. a. What are the betas of Stocks X and Y? b. What are the required rates of...
-
Refer to the information in Exercise 22-12. Assume that each of the companys divisions has a required rate of return of 7%. Compute residual income for each division. Data From Exercise 22-12 A food...
-
On July 1, a city issued, at par, $100 million in 6 percent, 20-year general obligation bonds. It established a debt service fund to account for resources set aside to pay interest and principal on...
-
By using your own words, describe Online Analytical Processing and what are the implementation techniques.?
-
A typical root beer contains 0.13% of a 75% H 3 PO 4 solution by mass. How many milligrams of phosphorus are contained in a 12 oz can of this root beer? Assume a solution density of 1.00 g/mL; also,...
-
An isotonic solution is described as 0.92% NaCl (mass/volume). Would this also be the required concentration for isotonic solutions of other salts, such as KCl, MgCl 2 , or MgSO 4 ? Explain.
-
Lorretta Klump, at the time a resident of Illinois, was injured in an automobile collision in which her vehicle was struck by a vehicle driven by Curt Eaves, also an Illinois resident. This incident...
-
Which of the following would not affect a nongovernment ONPO Statement of Activities? a. Depreciation expense. b. Expenditure of restricted contributions for the restricted purpose in the current...
-
Glenmore County Hospital's property, plant, and equipment (net of accumulated depreciation) consists of the following: Land \(\$ 500,000\) Buildings \(10,000,000\) Equipment (purchased from...
-
Unconditional promises to give that are restricted for the purpose of acquiring fixed assets should be recognized as contributions revenue by nongovernment VHWOs and ONPOs in the period(s) that: a....
-
The primary financial statement(s) that must be prepared by other not-for-profit organizations (ONPOs) do not include a: a. Balance Sheet. b. Statement of Cash Flows. c. Statement of Functional...
-
Which of the following fiduciary fund types, if any, does not have a measurement focus? a. Pension Trust Fund. b. Private-Purpose Trust Fund. c. Investment Trust Fund. d. Agency Fund. e. None of the...
-
Assume that oil begins to run out and that extraction becomes more expensive. Trace through the effects of this on the market for oil and the market for other fuels. Discuss.
-
For the following arrangements, discuss whether they are 'in substance' lease transactions, and thus fall under the ambit of IAS 17.
-
The Cambridge Opera Association has come up with a unique door prize for its December (2004) fund-raising ball: Twenty door prizes will be distributed, each one a ticket entitling the bearer to...
-
You are asked to value a large building in northern New Jersey. The valuation is needed for a bankruptcy settlement. Here are the facts: ? The settlement requires? that the building?s value equal the...
-
The manufacture of polysyllabic acid is a competitive industry. Most plants have an annual output of 100,000 tons. Operating costs are $.90 a ton, and the sales price is $1 a ton. A 100,000-ton plant...
-
During winter, the average temperature in Calgary is -5C, with a relative humidity of 75%, while the average temperature in Amsterdam is 5C, with a relative humidity of 90%. The skin temperature of a...
-
An elevator with a person in it is being pulled upwards by a massive cable. Identify all interaction forces (consistent with Newton's 3rd Law) in the system. Specify the object the force acts on, a...
-
Assuming a homogeneous, isotropic soil and a point load of 210 kN determine the increase in stress (kPa) at a depth 2.5 m below the point load at a radial distance 1.9 m. Provide your answer to 2...

Study smarter with the SolutionInn App


