Match the following compounds with the chemical properties in the next column. Write a chemical equation for
Question:
Match the following compounds with the chemical properties in the next column. Write a chemical equation for the reactions described in (a)-(d).
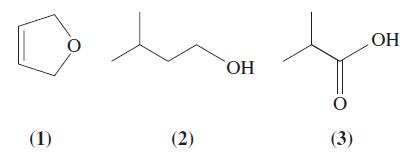
(a) Is easily oxidized;
(b) Decolorizes bromine water;
(c) Generates bubbles of gas when treated with Na2CO3(aq);
(d) Generates bubbles of gas when sodium metal is added.
Transcribed Image Text:
(1) (2) ОН 0 (3) ОН
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
a Is easily oxidized Aldehydes and ketones Chemical equation RC...View the full answer

Answered By

Asim farooq
I have done MS finance and expertise in the field of Accounting, finance, cost accounting, security analysis and portfolio management and management, MS office is at my fingertips, I want my client to take advantage of my practical knowledge. I have been mentoring my client on a freelancer website from last two years, Currently I am working in Telecom company as a financial analyst and before that working as an accountant with Pepsi for one year. I also join a nonprofit organization as a finance assistant to my job duties are making payment to client after tax calculation, I have started my professional career from teaching I was teaching to a master's level student for two years in the evening.
My Expert Service
Financial accounting, Financial management, Cost accounting, Human resource management, Business communication and report writing. Financial accounting : • Journal entries • Financial statements including balance sheet, Profit & Loss account, Cash flow statement • Adjustment entries • Ratio analysis • Accounting concepts • Single entry accounting • Double entry accounting • Bills of exchange • Bank reconciliation statements Cost accounting : • Budgeting • Job order costing • Process costing • Cost of goods sold Financial management : • Capital budgeting • Net Present Value (NPV) • Internal Rate of Return (IRR) • Payback period • Discounted cash flows • Financial analysis • Capital assets pricing model • Simple interest, Compound interest & annuities
4.40+
65+ Reviews
86+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Match the following compounds with the chemical properties given below. Write the structure of the products of the reactions described in (a) to (e). (a) Is easily oxidized; (b) Neutralizes NaOH(aq);...
-
a. Consider the chemical properties and physical properties of the halogens chlorine, bromine and iodine. Using these properties, predict the following about the other two halogens, fluorine and...
-
Organic chemists use a variety of methods to help them identify the functional groups in a molecule. In this chapter, we mentioned a few simple chemical ways to test for alkenes, alcohols, carboxylic...
-
Use the Ratio Test to determine the values of x 0 for which each series converges. 00 X 2k 2 k=1 k
-
You are at the student union having lunch with a friend who is attending law school. In the course of your conversation, you tell your friend that, in contrast to financial accounting or tax...
-
Filkins Farm Equipment needs to raise$4.5million for expansion, and it expects that five-year zero coupon bonds can be sold at a price of $567.44 for each $1,000 bond. a. How many $1,000 par value,...
-
Petitioner Atlantic Marine Company was a contractor located in Virginia. It entered into a subcontract with J-Crew Management, Inc., a Texas corporation, that contained a forum-selection clause...
-
Adjustment data for Oasis Health Care Inc. for January are as follows: 1. Insurance expired, $800. 2. Supplies on hand on January 31, $1,100. 3. Depreciation on building, $2,000. 4. Unearned rent...
-
Simplify: 6/8p 98p.
-
Match the following compounds with the chemical properties given below. Write a chemical equation for the reactions described in (a)-(d). (a) Forms an ester with ethanol (b) Absorbs H 2 in the...
-
How many elements of unsaturation are there in the molecule below? What is the molecular formula? Br CN
-
Discuss the following statement: Ratio analysis using financial statements is pointless. Only economic value added gives a true measure of the financial performance of a company.
-
If protons (H+) are reduced in a chemical reaction, what do they form? H (s) H(g) O H0 (1) HO (g)
-
using the ratio method w / the external calibration method, your standard of 3 . 0 1 0 ppm gave a response of 5 2 0 . 3 , with the unknown yielding a response of 4 8 1 . 4 . find the concentration of...
-
CaH2 + 2 HO -> Ca(OH)2 + 2H2 How many grams of calcium Hydride are needed to form 8.500g of hydrogen gas 354.31 g CaH2 177.15 g CaH2 2.10 g CaH 88.6 g CaH2 O
-
A fuel tank holds 2 2 . 3 gallons of gasoline. If the densityof gasoline is 0 . 8 2 0 6 g / mL , What is the mass inkilograms of gasoline in a full tank?
-
Which type of protein if HIF-1, which has increased activity in tumor cells in hypoxic conditions? O Antibody O Glycolysis enzyme O Transcription factor O Glucose transporter
-
Explain relationships between an exclusive agency contract and a power of attorney.
-
The bookkeeper for Riley, Inc., made the following errors: a. A cash purchase of supplies of $357 was recorded as a debit to Supplies for $375 and a credit to Cash of $375. b. A cash sale of $3,154...
-
The Shrieves Corporation has $10,000 that it plans to invest in marketable securities. It is choosing among AT&T bonds, which yield 7.5%, state of Florida muni bonds, which yield 5% (but are not...
-
The Moore Corporation has operating income (EBIT) of $750,000. The companys depreciation expense is $200,000. Moore is 100% equity financed, and it faces a 40% tax rate. What is the companys net...
-
The Berndt Corporation expects to have sales of $12 million. Costs other than depreciation are expected to be 75% of sales, and depreciation is expected to be $1.5 million. All sales revenues will be...
-
How does it works Flow and Pull ?what is Implementation of Diffie - Hellman Algorithm?
-
Assume that the law allows an MLB franchise owner to assign 50% of a team's purchase price to player contracts, and that they must be depreciated over one year. Freddy Moneybucks purchases the...
-
1. Does the concept of employment relationships as economic exchange significantly add to our understanding of reward strategies? 2. To what extent is it possible for managers to convince workers to...

Study smarter with the SolutionInn App


