Refer to Figure 11-25 to arrange the following species in the expected order of increasing (a) Bond
Question:
Refer to Figure 11-25 to arrange the following species in the expected order of increasing
(a) Bond length;
(b) Bond strength (energy): O2, O2+, O2–, O22–. State the basis of your expectation.
Figure 11-25
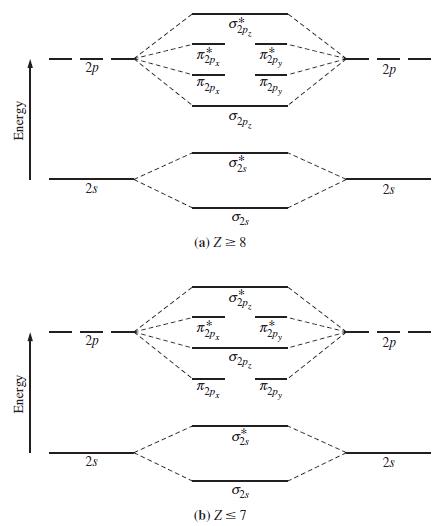
Transcribed Image Text:
Energy Energy 2p 28 2p 2s Tips โ ส Tips ni (a) Z = 8 สา 6. (b)Z≤7 TOY โ โ ลา 2p 2s 2p 28 T
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

General Chemistry Principles And Modern Applications
ISBN: 9780132931281
11th Edition
Authors: Ralph Petrucci, Jeffry Madura, F. Herring, Carey Bissonnette
Question Posted:
Students also viewed these Sciences questions
-
Arrange the following species in order of increasing stability: Li2, Li2+, Li2-2. Justify your choice with a molecular orbital energy level diagram?
-
Arrange the following species in order of increasing oxidation number of the sulfur atom: (a) H2S, (b) S8 (c) H2SO4, (d) S2-, (e) HS-, (f) SO2, (g) SO3.
-
(A) Refer only to the periodic table on the inside front cover, and arrange the following species in order of increasing size: Ti 2+ , V 3+ , Ca 2+ , Br - , and Sr 2+ . (B) Refer only to the periodic...
-
1. Should Sandy do exactly what the partner suggests, or risk losing the business for the firm, and perhaps her job, by suggesting a different approach? 2. What might be an alternative approach? 3....
-
Which of the following is not applicable to the internal audit function? a. Deter or catch employee fraud. b. Issue an opinion for investors regarding the reliability of the financial statements. c....
-
According to a recent survey by two United Nations agencies and a nongovernmental organization, two in every three women in the Indian capital of New Delhi are likely to face some form of sexual...
-
What is meant by the term fact pleading?
-
The 10-K of NIKE is reproduced in Appendix C. Required (a) What are the major sources of cash for the company, and what is it doing with that cash? (b) Were there any significant transactions in...
-
Kamron's Supply, Inc. provides cleaning supplies to the automobile detailing industry. It has two support departments providing support and operating functions. Support functions departments are...
-
Polonium is the only element known to crystallize in the simple cubic form. In this structure, the interatomic distance between a Po atom and each of its six nearest neighbors is 335 pm. Use this...
-
The following bond energies are given for 298 K: O 2 , 498; N 2 , 946; F 2 , 159; Cl 2 , 243; ClF, 251; OF (in OF 2 ), 213; ClO (in Cl 2 O), 205; and NF (in NF 3 ), 280 kJ mol 1 . Calculate f H at...
-
Refrigerant-134a is converted from a saturated liquid to a saturated vapor in a closed system using a reversible constant pressure process by transferring heat from a heat reservoir at 6C. From...
-
In a flow of air \(ho_{1}=1.35 \times 10^{5} \mathrm{~N} / \mathrm{m}^{2}, T_{1}=500 \mathrm{~K}\), and \(V_{1}=540 \mathrm{~m} / \mathrm{s}\). Heat transfer occurs in a constant-area duct until the...
-
Air flows at the rate of \(39.0 \mathrm{lbm} / \mathrm{sec}\) with a Mach number of 0.30 , a pressure of 50 psia, and a temperature of \(650^{\circ} \mathrm{R}\). The duct has a...
-
A certain container holds 50 monatomic particles with a combined energy of 200 units. 25 diatomic particles, with each particle having 2 units of energy, str then added and the system is allowed to...
-
Find the rate of change of entropy with respect to thermal energy of a system with an absolute temperature of \(100{ }^{\circ} \mathrm{C}\).
-
You poured freshly brewed hot tea into a cup. A thermometer indicates that its temperature is \(96.8^{\circ} \mathrm{C}\). Convert this temperature into the Fahrenheit and Kelvin scales.
-
Describe the auditors' responsibility for reviewing the footnotes and other material found in the annual report.
-
On July 1, 2011, Flashlight Corporation sold equipment it had recently purchased to an unaffiliated company for $480,000. The equipment had a book value on Flashlights books of $390,000 and a...
-
As its year-end approaches, it appears that Lopez Corporations net income will increase 10% this year. The president of Lopez Corporation, nervous that the stockholders might expect the company to...
-
Credit card usage in the United States is substantial. Many startup companies use credit cards as a way to help meet short-term financial needs. The most common forms of debt for startups are use of...
-
If your school has a subscription to the FASB Codification, to log in and prepare responses to the following. (a) How are receivables defined in the Codification? (b) What are the conditions under...
-
Oriole Railroad Co. is about to issue $260,000 of 10-year bonds paying an 10% interest rate, with interest payable annually. The discount rate for such securities is 8%. Click here to view the factor...
-
You have an air compressor capable of supplying 150 SCFM @ 150psi to a wind tunnel. The test section has the following maximumcross-sectional dimensions: Width = 0.5 in, Height = 2.0 in. Whatis the...
-
Air flows through a converging-diverging nozzle/diffuser.Assuming isentropic flow, air as an ideal gas, and constantspecific heats determine the state at several locations in thesystem. Note: The...

Study smarter with the SolutionInn App


